Key Insights
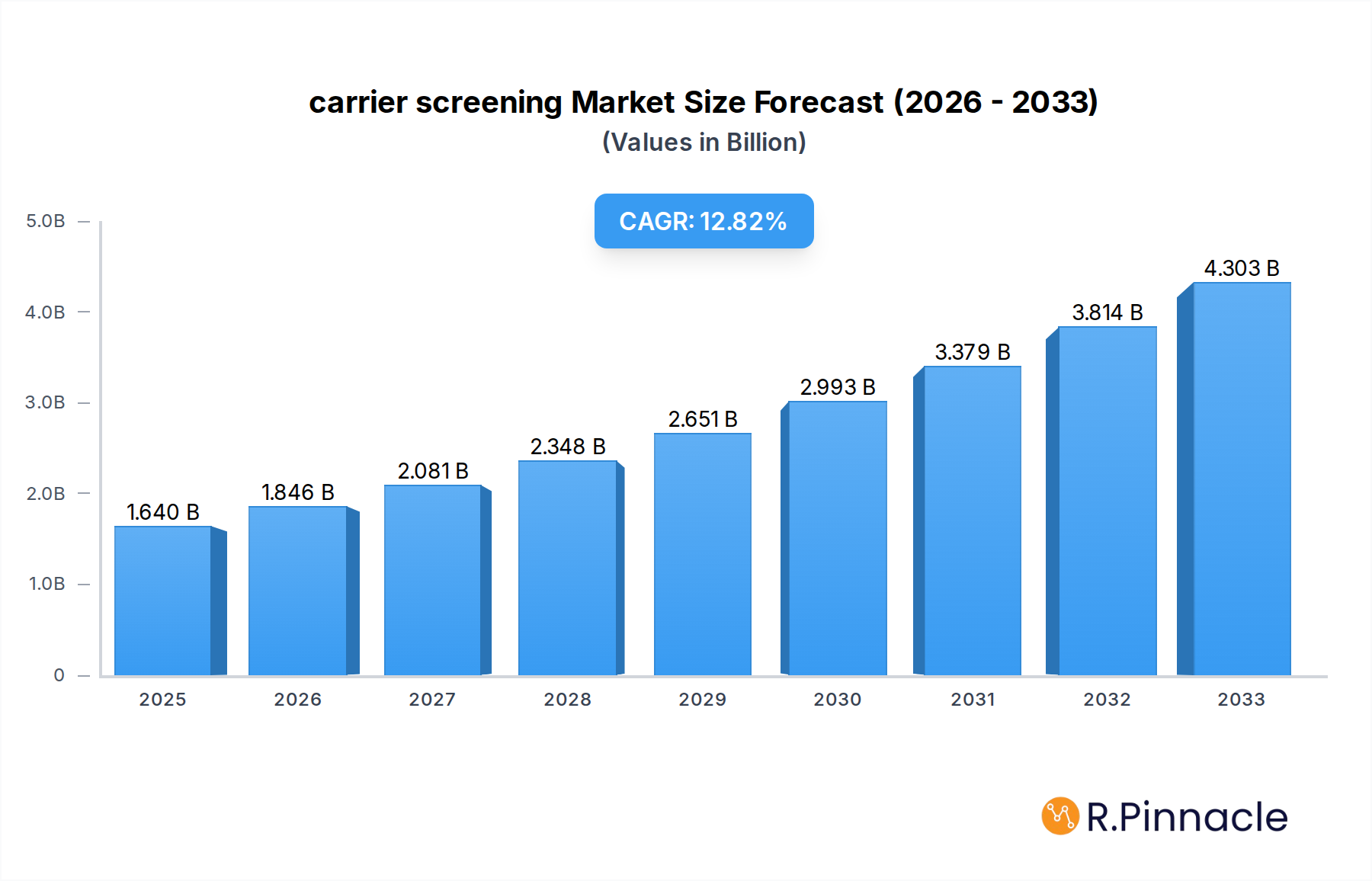
The global carrier screening market is poised for significant expansion, projected to reach USD 1.64 billion in 2025 and experience a robust CAGR of 12.55% through 2033. This impressive growth is fueled by an increasing awareness of genetic disorders and the proactive desire among prospective parents to understand their reproductive risks. The market is driven by advancements in molecular diagnostics, leading to more accurate, accessible, and affordable screening tests. Furthermore, the rising prevalence of inherited diseases, coupled with growing governmental initiatives and healthcare provider recommendations for routine carrier screening, especially in preconception and prenatal settings, are major catalysts. The expanding integration of carrier screening into standard reproductive healthcare practices across diverse medical facilities, including hospitals, clinics, and ambulatory surgical centers, is also contributing to its widespread adoption.
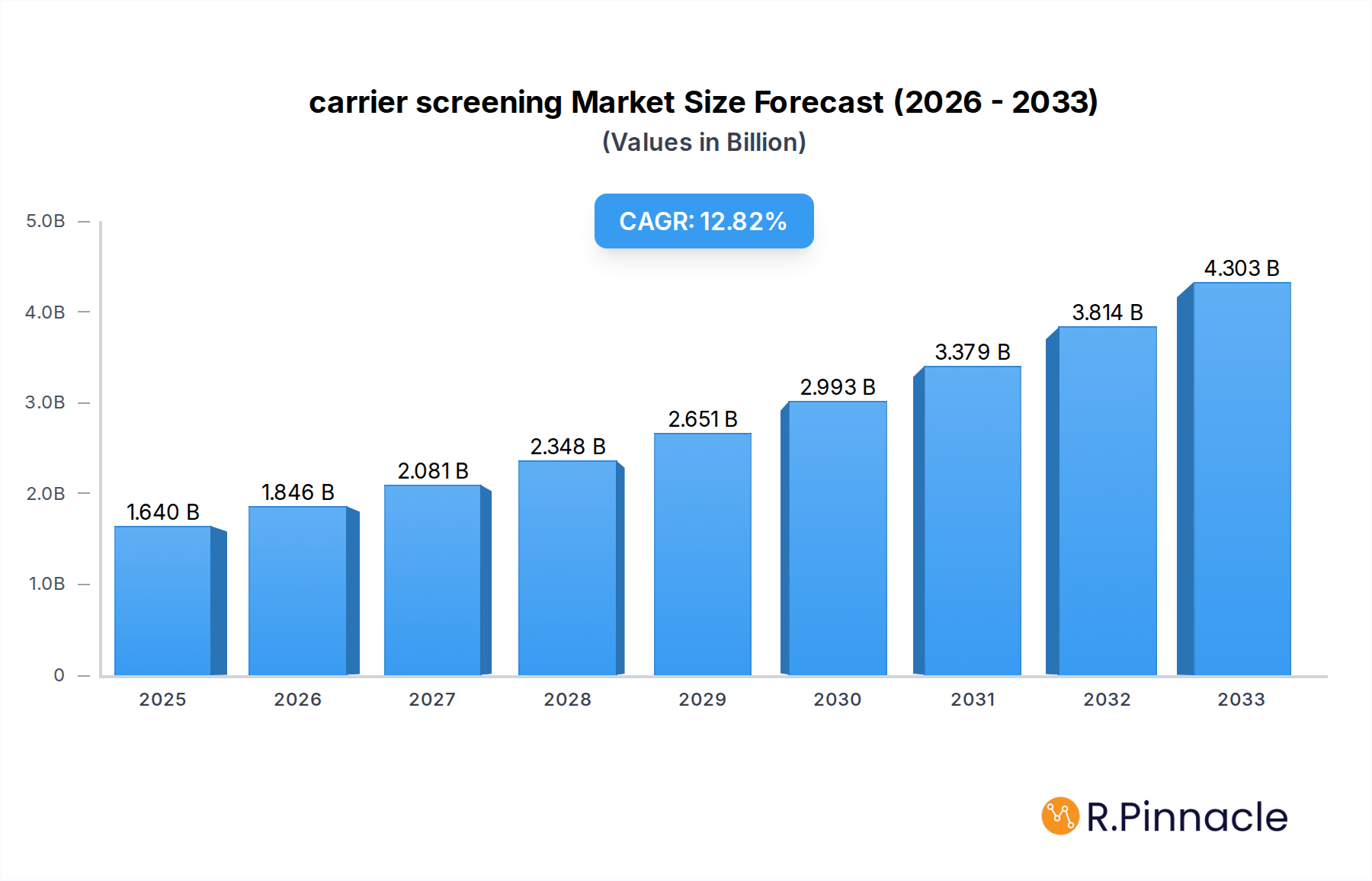
carrier screening Market Size (In Billion)

The market is segmented into molecular screening tests and biochemical screening tests, with molecular methods dominating due to their superior sensitivity and specificity in detecting a broader range of genetic mutations. Key market players such as Abbott Laboratories, Roche, Thermo Fisher Scientific, Illumina, and LabCorp are at the forefront of innovation, investing heavily in research and development to enhance test capabilities and broaden the scope of detectable conditions. Emerging trends like the integration of artificial intelligence for data analysis and personalized risk assessment, alongside the growing demand for direct-to-consumer genetic testing services, are shaping the future landscape. However, potential restraints such as the cost of advanced genetic sequencing technologies and varying regulatory frameworks across different regions could present challenges. Despite these, the overall outlook for the carrier screening market remains exceptionally strong, driven by a fundamental shift towards preventative healthcare and informed reproductive choices.
carrier screening Company Market Share

Here's an SEO-optimized, reader-centric report description for carrier screening, designed for industry professionals and leveraging high-ranking keywords.
Carrier Screening Market Report: Innovations, Dynamics, and Future Outlook (2019-2033)
This comprehensive report delves into the rapidly evolving global carrier screening market, providing deep insights into market structure, innovation trends, growth drivers, challenges, and future projections. Analyzing data from 2019 through 2033, with a base and estimated year of 2025, this study is essential for stakeholders seeking to understand the strategic landscape, competitive dynamics, and technological advancements shaping this critical segment of diagnostic healthcare. The report covers key applications in Hospitals, Clinics, and Ambulatory Surgical Centers, and segments by Molecular Screening Test and Biochemical Screening Test types.
Carrier Screening Market Structure & Innovation Trends
The global carrier screening market, projected to reach billions in value by 2033, exhibits a dynamic structure characterized by a blend of established industry giants and emerging innovators. Market concentration is influenced by significant R&D investments, stringent regulatory approvals, and strategic mergers and acquisitions. Innovation is primarily driven by advancements in next-generation sequencing (NGS) and polymerase chain reaction (PCR) technologies, enabling more comprehensive and accurate genetic profiling. Key innovation drivers include the demand for personalized medicine, increased awareness of genetic disorders, and the expanding utility of carrier screening beyond reproductive health. Regulatory frameworks, overseen by bodies like the FDA, play a crucial role in ensuring test accuracy and patient safety, acting as both enablers and potential barriers to market entry. Product substitutes, such as traditional genetic counseling, are increasingly being complemented by advanced molecular screening tests. End-user demographics are shifting towards a younger, more health-conscious population seeking proactive genetic risk assessment. M&A activities, valued in the billions, are a prominent feature, consolidating market share and expanding technological portfolios. For instance, recent M&A deals have focused on integrating diagnostic platforms and enhancing data analytics capabilities.
Carrier Screening Market Dynamics & Trends
The carrier screening market is poised for substantial growth, driven by a confluence of technological breakthroughs, evolving consumer preferences, and expanding healthcare access. The global market is witnessing an impressive Compound Annual Growth Rate (CAGR) of xx% from 2025 to 2033, indicative of robust expansion. This growth is fueled by increasing awareness among prospective parents regarding inherited genetic conditions and the desire to ensure the health of future generations. The penetration of advanced genetic testing technologies, particularly Molecular Screening Tests, is accelerating rapidly. Technological disruptions, such as the integration of artificial intelligence (AI) and machine learning (ML) in data analysis and interpretation, are enhancing diagnostic accuracy and offering predictive insights. Consumer preferences are increasingly leaning towards non-invasive prenatal testing (NIPT) and comprehensive pan-ethnic screening panels that cover a wider range of genetic disorders. The competitive landscape is characterized by intense innovation, with companies like Roche, Thermo Fisher Scientific, and Illumina investing heavily in R&D to develop more sensitive and cost-effective screening solutions. Market penetration is also being boosted by government initiatives promoting genetic screening programs and the growing adoption of these tests in routine clinical practice, particularly within Hospitals and specialized Clinics. The shift towards direct-to-consumer (DTC) genetic testing services, offered by companies like 23andMe, is further broadening market reach, though regulatory scrutiny remains a significant factor. The expanding application of carrier screening in areas beyond reproductive health, such as oncology and rare disease identification, is also a key growth accelerant.
Dominant Regions & Segments in Carrier Screening
North America currently stands as the dominant region in the global carrier screening market, with an estimated market share of xx billion dollars in 2025. This leadership is attributed to a robust healthcare infrastructure, high disposable incomes, advanced technological adoption, and strong government support for genetic research and diagnostics. The United States, in particular, plays a pivotal role, driven by the presence of leading market players and a proactive approach to adopting innovative healthcare solutions.
Within North America, the Molecular Screening Test segment is experiencing exceptional growth, outpacing Biochemical Screening Tests. This dominance is fueled by the superior sensitivity, specificity, and comprehensiveness of molecular techniques like Next-Generation Sequencing (NGS).
Key Drivers for Dominance in North America:
- Economic Policies: Favorable reimbursement policies and insurance coverage for genetic testing services significantly boost market adoption.
- Infrastructure: Well-established diagnostic laboratory networks and advanced healthcare facilities support widespread implementation.
- Research & Development: High levels of investment in genetic research by both public and private entities lead to continuous innovation.
In terms of application, Clinics are emerging as a significant growth area within the carrier screening landscape. While Hospitals traditionally lead in diagnostic volumes, the increasing preference for specialized, outpatient genetic counseling and testing services is driving growth in clinic settings. Ambulatory Surgical Centers are also showing steady growth as more screening procedures are integrated into outpatient care pathways.
The Molecular Screening Test segment is projected to maintain its lead throughout the forecast period (2025-2033). This is due to its ability to detect a broader spectrum of genetic mutations with higher accuracy, addressing the growing demand for pan-ethnic screening panels and comprehensive risk assessments. Companies like Illumina and Thermo Fisher Scientific are at the forefront of molecular diagnostic innovation, continually refining technologies to improve speed, accuracy, and cost-effectiveness.
Carrier Screening Product Innovations
Product innovation in carrier screening is primarily centered on enhancing the scope and accuracy of genetic analysis. Companies are developing advanced molecular screening tests that leverage Next-Generation Sequencing (NGS) and CRISPR-based technologies to detect a wider array of genetic disorders with unparalleled precision. These innovations offer significant competitive advantages by providing comprehensive pan-ethnic panels, enabling personalized risk assessments, and reducing the likelihood of false negatives. The integration of artificial intelligence in data interpretation further refines diagnostic capabilities, offering clinicians deeper insights into patient genetic profiles. These developments are crucial for meeting the growing demand for proactive reproductive health and rare disease diagnosis.
Report Scope & Segmentation Analysis
This report comprehensively segments the carrier screening market across key categories to provide granular insights into market dynamics. The Application segmentation includes Hospitals, Clinics, and Ambulatory Surgical Centers. Each of these segments is analyzed for market size, growth projections, and competitive dynamics, with Clinics expected to demonstrate significant growth in the coming years due to increasing demand for specialized outpatient genetic services. The Type segmentation distinguishes between Molecular Screening Tests and Biochemical Screening Tests. The Molecular Screening Test segment is anticipated to dominate the market, driven by technological advancements and its ability to offer more comprehensive genetic analysis, with projected market sizes reaching billions by 2033 and robust CAGR.
Key Drivers of Carrier Screening Growth
The carrier screening market is experiencing robust growth propelled by several key factors. Technological advancements, particularly in molecular diagnostics such as Next-Generation Sequencing (NGS) and CRISPR, are enabling more accurate, comprehensive, and cost-effective testing for a wider range of genetic disorders. This is complemented by increasing public awareness regarding inherited diseases and the importance of genetic testing for family planning and preventative healthcare. Favorable regulatory landscapes in many regions, coupled with expanding insurance coverage and reimbursement policies for genetic screening, are making these tests more accessible. Furthermore, the growing trend towards personalized medicine and the demand for proactive health management are significant catalysts.
Challenges in the Carrier Screening Sector
Despite its promising growth trajectory, the carrier screening sector faces several challenges. Regulatory hurdles, including the complex approval processes for new diagnostic tests and varying regulations across different countries, can impede market entry and product commercialization. High costs associated with advanced genetic testing technologies, while decreasing, can still be a barrier to widespread adoption, particularly in resource-limited settings. Ethical considerations surrounding genetic data privacy, potential for genetic discrimination, and the psychological impact of genetic risk information require careful management. Furthermore, ensuring equitable access to carrier screening across diverse socioeconomic and ethnic populations remains a significant challenge, alongside the need for continuous education for healthcare providers and the public.
Emerging Opportunities in Carrier Screening
The carrier screening market is ripe with emerging opportunities. The development and adoption of pan-ethnic screening panels that cater to diverse genetic backgrounds present a significant avenue for growth, addressing limitations of older, ethnicity-specific tests. Expansion into non-reproductive applications, such as newborn screening, rare disease diagnosis, and pharmacogenomics, offers new market segments. The increasing integration of artificial intelligence (AI) and machine learning (ML) for improved data interpretation, risk prediction, and diagnostic accuracy is a major technological frontier. Furthermore, the growing demand for direct-to-consumer (DTC) genetic testing services, coupled with partnerships between DTC providers and clinical laboratories, opens up new distribution channels and consumer engagement models.
Leading Players in the Carrier Screening Market
- Abbott Laboratories
- Roche
- Thermo Fisher Scientific
- 23andMe
- Danaher
- Illumina
- Luminex
- LabCorp
- Myriad Genetics
- Autogenomics
Key Developments in Carrier Screening Industry
- 2023 December: Illumina launches new NGS-based carrier screening panel with expanded gene coverage, enhancing diagnostic capabilities.
- 2023 November: Thermo Fisher Scientific expands its molecular diagnostic portfolio with a novel biochemical screening assay for rare metabolic disorders.
- 2023 October: Roche announces strategic partnership with a leading genomics company to integrate advanced AI for carrier screening data analysis.
- 2023 September: 23andMe receives expanded FDA clearance for its carrier status reports, covering a broader range of inherited conditions.
- 2023 July: Danaher invests heavily in expanding its genetic testing laboratory capacity to meet growing demand.
- 2023 April: Abbott Laboratories introduces a new point-of-care carrier screening solution for increased accessibility.
- 2023 February: Luminex acquires a biotechnology firm specializing in molecular diagnostics for rare genetic diseases.
- 2022 November: LabCorp enhances its carrier screening services with advanced bioinformatics and genetic counseling support.
- 2022 August: Myriad Genetics expands its genetic testing offerings with a focus on hereditary cancer predisposition.
- 2022 May: Autogenomics receives regulatory approval for its multiplex molecular diagnostic platform for carrier screening.
Future Outlook for Carrier Screening Market
The future outlook for the carrier screening market is exceptionally bright, characterized by sustained growth and transformative innovation. The increasing global emphasis on preventative healthcare, coupled with advancements in genetic technologies, will continue to drive market expansion. We anticipate a significant rise in the adoption of comprehensive pan-ethnic carrier screening panels and the further integration of AI and ML into diagnostic workflows, enhancing precision and accessibility. The expansion of carrier screening into non-reproductive applications and emerging markets will unlock substantial new revenue streams. Strategic collaborations and a focus on personalized genetic solutions will be key to navigating the competitive landscape and capitalizing on the evolving demands of patients and healthcare providers.
carrier screening Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Ambulatory Surgical Centers
-
2. Types
- 2.1. Molecular Screening Test
- 2.2. Biochemical Screening Test
carrier screening Segmentation By Geography
- 1. CA
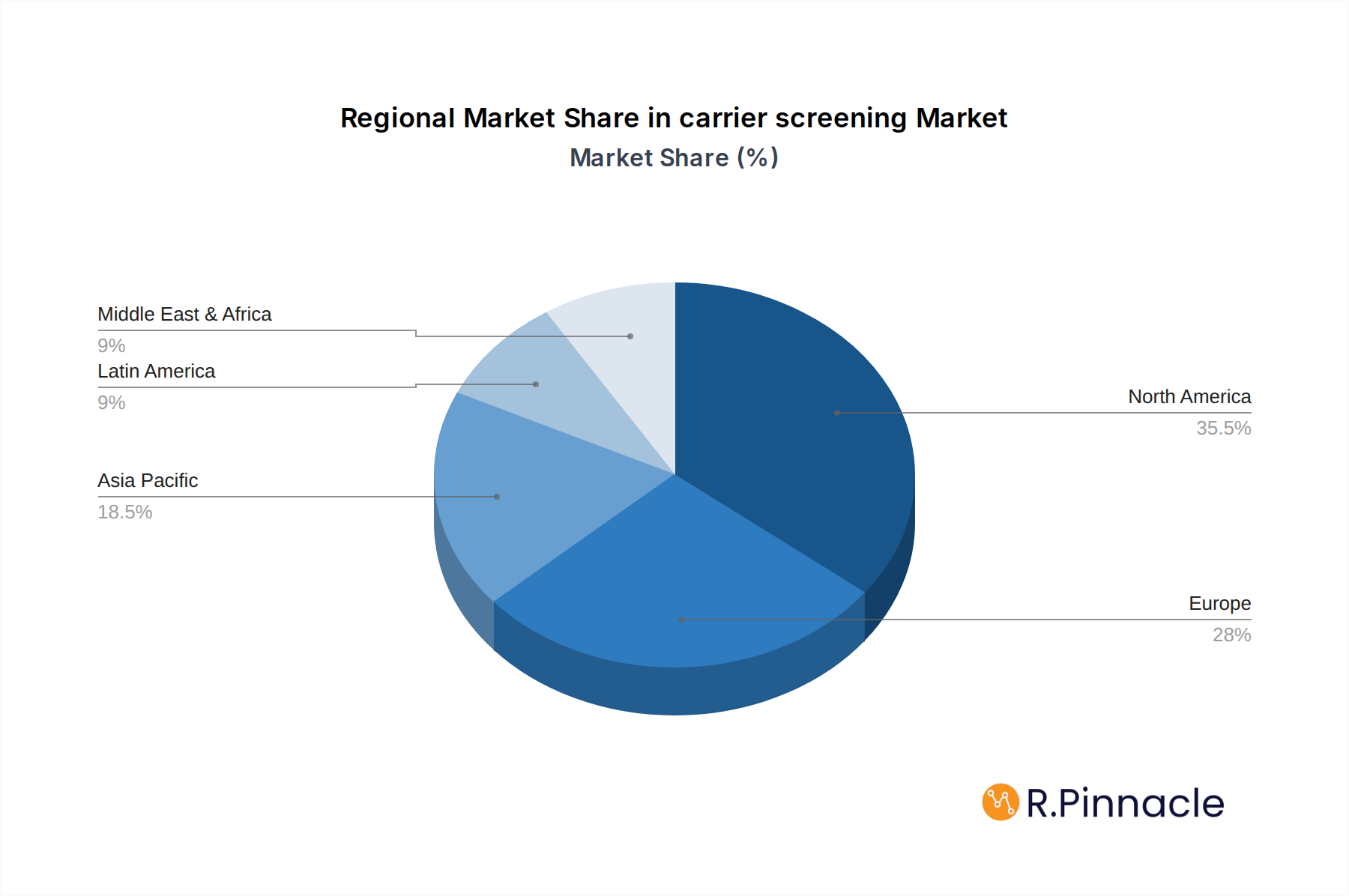
carrier screening Regional Market Share

Geographic Coverage of carrier screening
carrier screening REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.55% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. carrier screening Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Ambulatory Surgical Centers
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Molecular Screening Test
- 5.2.2. Biochemical Screening Test
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Abbott Laboratories
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Roche
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Thermo Fisher Scientific
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 23andMe
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Danaher
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Illumina
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Luminex
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 LabCorp
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Myriad Genetics
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Autogenomics
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.1 Abbott Laboratories
List of Figures
- Figure 1: carrier screening Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: carrier screening Share (%) by Company 2025
List of Tables
- Table 1: carrier screening Revenue billion Forecast, by Application 2020 & 2033
- Table 2: carrier screening Revenue billion Forecast, by Types 2020 & 2033
- Table 3: carrier screening Revenue billion Forecast, by Region 2020 & 2033
- Table 4: carrier screening Revenue billion Forecast, by Application 2020 & 2033
- Table 5: carrier screening Revenue billion Forecast, by Types 2020 & 2033
- Table 6: carrier screening Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the carrier screening?
The projected CAGR is approximately 12.55%.
2. Which companies are prominent players in the carrier screening?
Key companies in the market include Abbott Laboratories, Roche, Thermo Fisher Scientific, 23andMe, Danaher, Illumina, Luminex, LabCorp, Myriad Genetics, Autogenomics.
3. What are the main segments of the carrier screening?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.64 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "carrier screening," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the carrier screening report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the carrier screening?
To stay informed about further developments, trends, and reports in the carrier screening, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
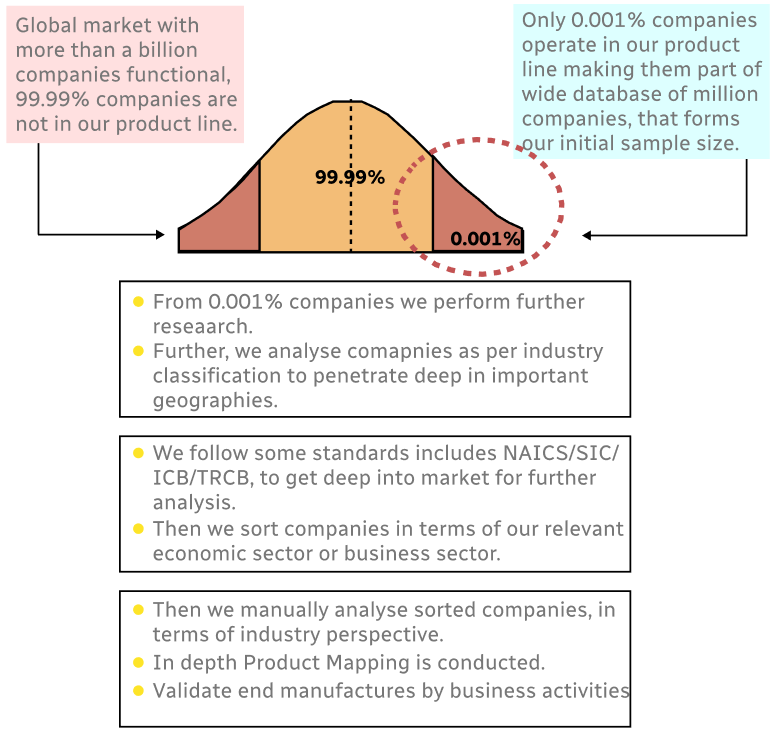
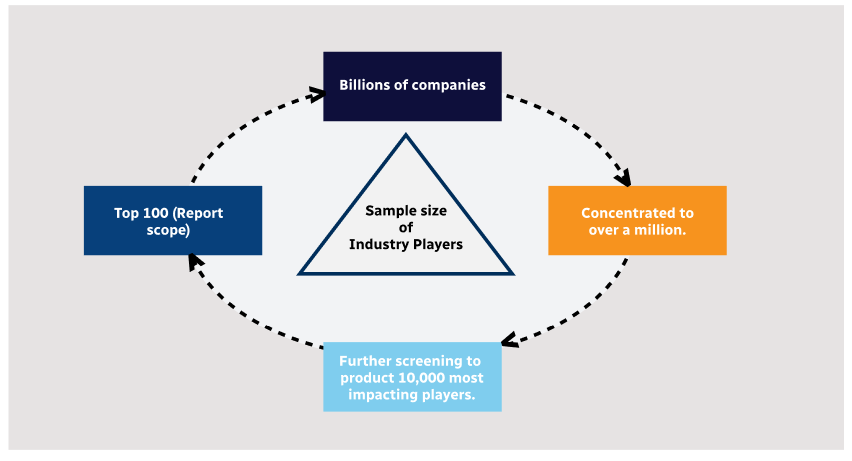
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
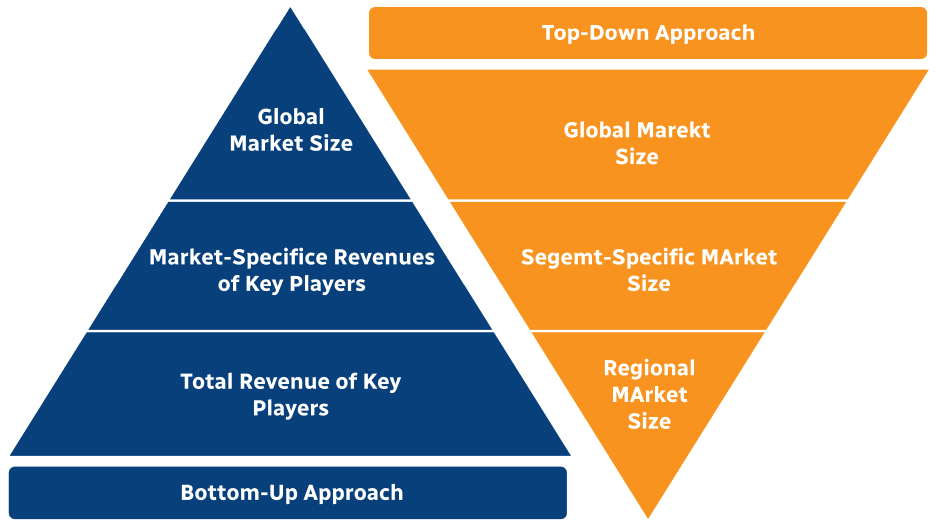
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
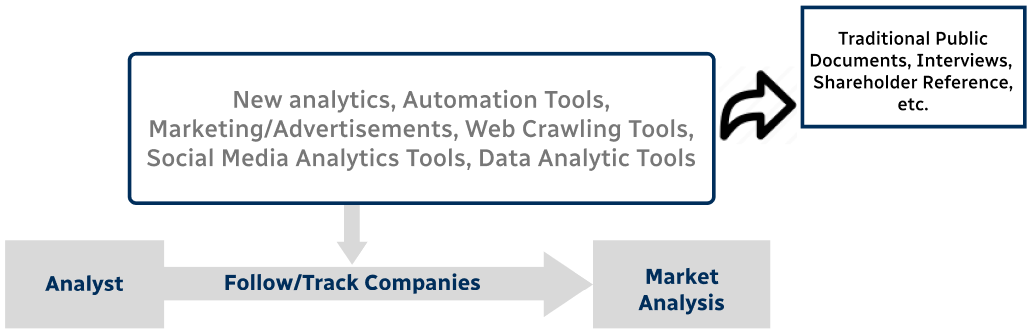
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


