Key Insights
The global CD48 antibody market is poised for robust expansion, projected to reach USD 12.72 billion in 2025 with a compelling Compound Annual Growth Rate (CAGR) of 6.8% over the forecast period of 2025-2033. This significant growth is primarily fueled by the escalating demand for advanced diagnostic tools and therapeutic interventions in oncology and immunology. The increasing prevalence of autoimmune diseases and infectious diseases, coupled with a growing emphasis on personalized medicine, are key drivers propelling the adoption of CD48 antibodies. Furthermore, continuous advancements in antibody engineering and a burgeoning pipeline of CD48-targeted therapies are expected to unlock new market opportunities, particularly in regions with substantial healthcare infrastructure and R&D investments.
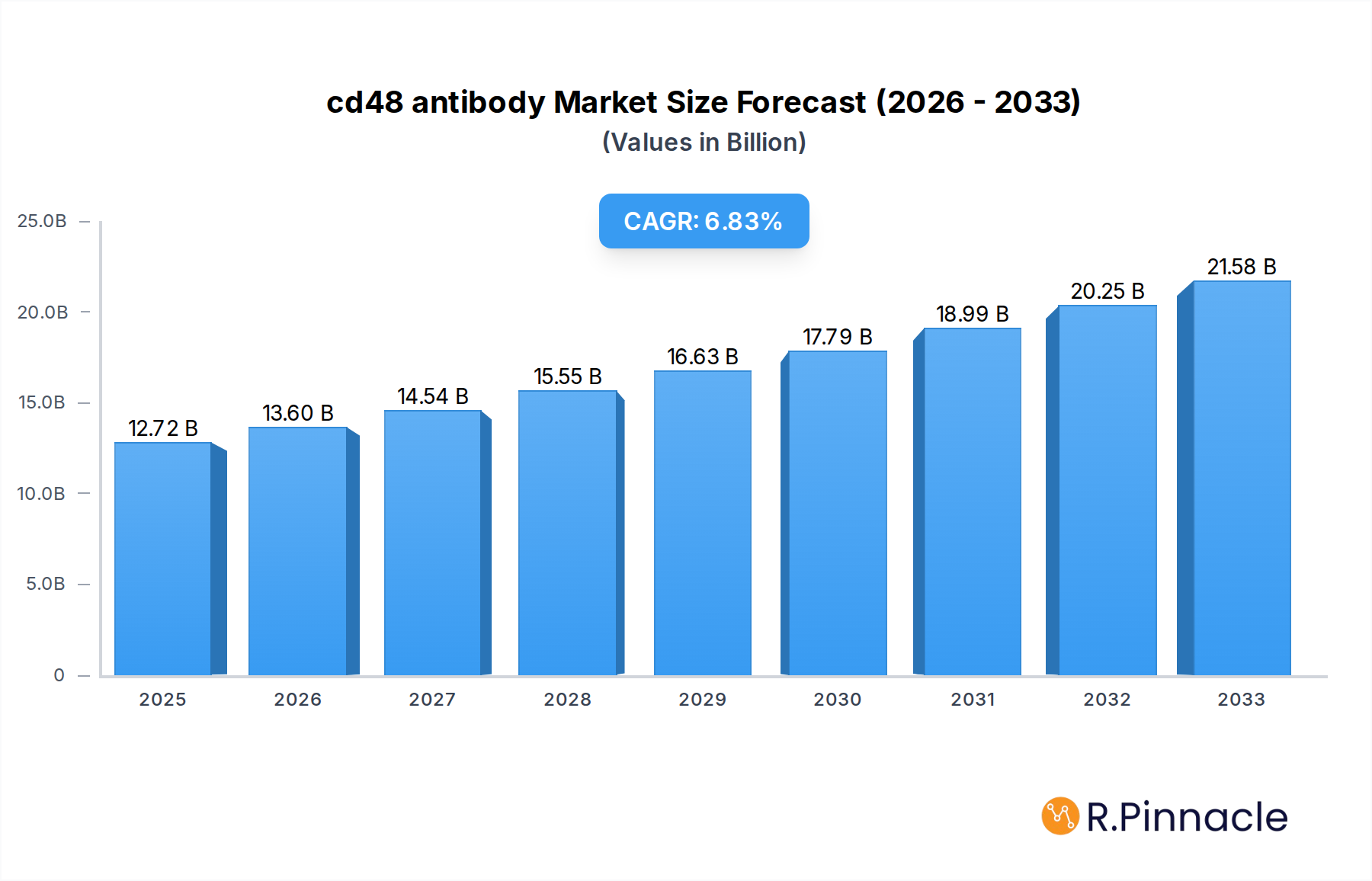
cd48 antibody Market Size (In Billion)

The market is segmented across various applications, including Flow Cytometry, ELISA, Western Blot, Immunoprecipitation, and Immunofluorescence, each contributing to the overall market dynamics. Monoclonal antibodies are expected to dominate the market due to their high specificity and efficacy, though polyclonal antibodies will continue to hold a significant share owing to their cost-effectiveness and broad applicability. North America is anticipated to lead the market, driven by a well-established research ecosystem and high healthcare expenditure. Asia Pacific, with its rapidly expanding biopharmaceutical sector and increasing R&D activities, is expected to exhibit the fastest growth rate. Key industry players are actively engaged in strategic collaborations and product launches to capitalize on these expanding opportunities and address unmet clinical needs.
cd48 antibody Company Market Share

This in-depth report provides a comprehensive analysis of the global CD48 antibody market, offering strategic insights and actionable intelligence for industry stakeholders. Leveraging advanced analytical techniques and extensive market data, this report explores market structure, dynamics, key growth drivers, challenges, opportunities, and the competitive landscape from 2019 to 2033, with a base year of 2025.
CD48 Antibody Market Structure & Innovation Trends
The global CD48 antibody market exhibits a moderate concentration, with a significant presence of established biotechnology companies and a growing number of specialized players. Innovation is primarily driven by advancements in antibody engineering, improved assay development, and a deeper understanding of CD48's role in various biological processes. Regulatory frameworks, particularly concerning quality control and biosafety, play a crucial role in shaping market entry and product approval. Substitutes for CD48 antibodies are currently limited, as they offer highly specific targeting. End-user demographics are primarily academic research institutions, pharmaceutical and biopharmaceutical companies, and contract research organizations. Mergers and acquisitions (M&A) activities, valued in the hundreds of billions, are observed as companies seek to expand their portfolios and gain market share. For instance, recent M&A deals in the broader immunology reagent sector have seen valuations in the tens of billions, reflecting the strategic importance of these technologies. Market share analysis reveals a dynamic landscape where innovation and strategic partnerships dictate leadership positions.
CD48 Antibody Market Dynamics & Trends
The CD48 antibody market is poised for significant expansion, driven by the escalating demand for precise diagnostic tools and targeted therapeutic interventions. The global market is projected to experience a Compound Annual Growth Rate (CAGR) of approximately 12.50% from 2025 to 2033, reaching an estimated market valuation of over $50 billion by the end of the forecast period. Market penetration is currently estimated at around 35% but is expected to grow substantially as awareness and applications broaden.
Key Growth Drivers:
- Advancements in Immunotherapy: The burgeoning field of immunotherapy, particularly in oncology and autoimmune diseases, relies heavily on understanding immune cell interactions. CD48, a crucial modulator of T cell activation and differentiation, is a prime target for research and therapeutic development, fueling demand for high-quality CD48 antibodies.
- Increasing Prevalence of Chronic Diseases: The rising incidence of chronic diseases, including various cancers, autoimmune disorders, and infectious diseases, necessitates advanced research tools for diagnosis, prognosis, and drug discovery. CD48 antibodies are indispensable in unraveling disease mechanisms and identifying novel therapeutic targets.
- Technological Innovations in Research Tools: Continuous advancements in antibody production techniques, such as recombinant DNA technology and single B cell cloning, are leading to the development of highly specific, sensitive, and reliable CD48 antibodies. This enhances experimental outcomes and accelerates research timelines.
- Growing Investments in Life Sciences R&D: Global investments in life sciences research and development are on an upward trajectory, with significant funding allocated to immunology and cancer research. This surge in R&D expenditure directly translates to increased demand for reagents like CD48 antibodies.
- Expansion of Diagnostics Market: The growing emphasis on early disease detection and personalized medicine is driving the expansion of the in-vitro diagnostics market. CD48 antibodies are finding increasing applications in diagnostic assays for various immunological conditions.
- Shift Towards Biologics and Precision Medicine: The pharmaceutical industry's increasing focus on developing biologics and adopting precision medicine approaches necessitates a deeper understanding of cellular interactions. CD48 plays a key role in these interactions, making CD48 antibodies vital for drug development and validation.
Technological Disruptions:
- CRISPR-Cas9 Gene Editing: While not directly producing antibodies, advancements in gene editing technologies are facilitating the creation of more relevant cellular models for studying CD48 function, thereby increasing the demand for well-characterized antibodies to validate these models.
- High-Throughput Screening (HTS): HTS platforms are increasingly being employed in drug discovery. The development of robust assays utilizing CD48 antibodies is crucial for identifying novel drug candidates that modulate CD48 pathways.
- Single-Cell Analysis Technologies: Emerging single-cell technologies allow for the dissection of cellular heterogeneity. CD48 antibodies are essential for identifying and characterizing specific cell populations expressing CD48 at the single-cell level, providing unprecedented insights into immune responses.
Consumer Preferences:
- High Specificity and Purity: Researchers and diagnostic developers prioritize antibodies with exceptional specificity to CD48, minimizing off-target effects and ensuring reliable experimental results. High purity levels are also paramount for sensitive assays.
- Reproducibility and Lot-to-Lot Consistency: Consistent performance across different experiments and between different batches is a critical factor influencing purchasing decisions. Manufacturers offering validated and well-characterized antibodies gain a competitive edge.
- Comprehensive Validation Data: Users increasingly seek antibodies with extensive validation data across multiple applications, including Western Blot, Flow Cytometry, and Immunofluorescence, to ensure their suitability for specific research needs.
- Technical Support and Application Notes: Access to responsive technical support and detailed application notes can significantly influence purchasing decisions, especially for complex research projects.
Competitive Dynamics:
The competitive landscape is characterized by a mix of large, established life science reagent providers and smaller, specialized antibody manufacturers. Competition is driven by product quality, innovation, pricing, and the breadth of the product portfolio. Strategic partnerships, licensing agreements, and the development of proprietary antibody technologies are key strategies employed by leading players to maintain and enhance their market position. The market is dynamic, with constant innovation and new product introductions.
Dominant Regions & Segments in CD48 Antibody
North America is currently the dominant region in the global CD48 antibody market, driven by substantial investments in life sciences research, a robust biopharmaceutical industry, and a high prevalence of research institutions and diagnostic laboratories. The United States, in particular, accounts for a significant share of the market due to its advanced healthcare infrastructure, strong government funding for scientific research, and a thriving ecosystem of biotechnology companies.
Key Drivers of Regional Dominance in North America:
- R&D Expenditure: The United States leads global R&D spending in the life sciences, with a significant portion allocated to immunology, cancer research, and drug discovery – areas where CD48 plays a crucial role. This high level of investment directly translates to a greater demand for CD48 antibodies.
- Biotechnology Hubs: The presence of major biotechnology hubs in regions like Boston, San Francisco Bay Area, and San Diego fosters innovation and collaboration, accelerating the adoption of new research tools and technologies, including advanced CD48 antibodies.
- Regulatory Environment: While stringent, the regulatory environment in North America, particularly the FDA's oversight, ensures the quality and reliability of biological reagents, which instills confidence among researchers and diagnostic developers.
- Academic Excellence: Leading universities and research institutions in North America are at the forefront of immunological research, driving demand for high-quality CD48 antibodies for basic science and translational research.
- Prevalence of Chronic Diseases: The high incidence of chronic diseases in North America, such as cancer and autoimmune disorders, necessitates continuous research and development, further fueling the demand for CD48 antibodies for both research and potential diagnostic applications.
Dominant Segments:
- Application: Flow Cytometry: Flow cytometry is a leading application for CD48 antibodies due to its ability to analyze cell surface markers on individual cells. This technique is invaluable for immunophenotyping, immune cell subset identification, and tracking immune cell activation in both research and clinical settings. The precision and sensitivity offered by CD48 antibodies in flow cytometry are critical for unraveling complex immune responses.
- Type: Monoclonal Antibody: Monoclonal antibodies represent the dominant type of CD48 antibody in the market. Their high specificity, consistency, and ability to recognize a single epitope on the CD48 protein make them indispensable for precise research applications and potential therapeutic development. The development of highly characterized monoclonal CD48 antibodies has been a significant enabler of progress in immunology.
The dominance of Flow Cytometry and Monoclonal Antibodies is further supported by the fact that these segments align perfectly with the current research trends in immunology, particularly in understanding immune cell interactions and developing targeted therapies. The growth in these segments is expected to continue, driven by ongoing technological advancements and the increasing complexity of immunological research questions being addressed. The estimated market share for Flow Cytometry applications is projected to be around 30 billion, with Monoclonal Antibodies capturing a similar share.
CD48 Antibody Product Innovations
Recent product innovations in the CD48 antibody market focus on enhancing specificity, improving sensitivity, and expanding compatibility across diverse research applications. Key developments include the introduction of highly validated monoclonal antibodies for multiplexing assays, enabling simultaneous detection of multiple targets. Furthermore, researchers are developing recombinant CD48 antibodies for increased batch-to-batch consistency and reduced immunogenicity, paving the way for potential therapeutic applications. Innovations also include conjugation of CD48 antibodies with advanced fluorophores for enhanced signal detection in microscopy and flow cytometry, and development of antibodies optimized for specific research areas like T-cell activation and immune checkpoint modulation. These advancements aim to provide researchers with more reliable and efficient tools, accelerating discoveries in immunology and related fields, with a projected market impact of over 15 billion in the next decade.
Report Scope & Segmentation Analysis
This report encompasses a comprehensive analysis of the global CD48 antibody market, segmented by key applications and antibody types. The study period spans from 2019 to 2033, with a base year of 2025.
- Application: The market is segmented into Flow Cytometry, ELISA, Western Blot, Immunoprecipitation, Immunofluorescence, and Others. Flow Cytometry is anticipated to maintain its leading position due to its extensive use in immunophenotyping and cell analysis, with projected growth exceeding 10 billion. ELISA is expected to witness steady growth in diagnostic applications, projected to reach over 5 billion. Western Blot and Immunofluorescence are crucial for protein detection and localization, contributing billions to the market.
- Type: The market is further segmented into Monoclonal Antibody and Polyclonal Antibody. Monoclonal antibodies are projected to dominate the market, driven by their high specificity and consistency, with an estimated market value of over 40 billion. Polyclonal antibodies, while offering broader epitope coverage, are expected to represent a smaller but stable segment, valued in the billions.
The detailed segmentation analysis indicates a robust and growing market, with specific segments exhibiting strong growth trajectories driven by their critical roles in scientific research and diagnostics.
Key Drivers of CD48 Antibody Growth
The growth of the CD48 antibody market is propelled by several interconnected factors:
- Advancements in Immunology Research: A deeper understanding of immune cell function, cell-cell interactions, and the role of CD48 in immune responses is continuously expanding the research applications for CD48 antibodies, particularly in areas like autoimmunity, transplantation, and cancer immunology.
- Expanding Applications in Diagnostics: The increasing need for precise diagnostic tools for various immunological disorders and cancers is driving the development and adoption of CD48 antibodies in diagnostic kits and assays.
- Growth of the Biopharmaceutical Sector: The burgeoning biopharmaceutical industry, with its focus on developing targeted therapies and biologics, requires reliable reagents like CD48 antibodies for drug discovery, preclinical testing, and validation.
- Technological Progress in Antibody Production: Innovations in antibody engineering and recombinant production technologies are leading to the availability of higher quality, more specific, and consistent CD48 antibodies, further fueling market growth.
- Government Funding and Initiatives: Increased government funding for life sciences research and specific initiatives focused on disease eradication and understanding the immune system directly impact the demand for research reagents like CD48 antibodies.
Challenges in the CD48 Antibody Sector
Despite its promising growth, the CD48 antibody sector faces several challenges:
- Stringent Quality Control and Regulatory Hurdles: Ensuring the consistent quality, purity, and specificity of antibodies requires rigorous quality control measures and adherence to evolving regulatory standards, which can be time-consuming and costly.
- High Cost of Production: The development and production of high-quality, validated antibodies, especially monoclonal antibodies, can be expensive, impacting affordability for some research institutions.
- Competition from Alternative Technologies: While CD48 antibodies are highly specific, ongoing advancements in alternative research technologies, such as CRISPR-based functional screening, could potentially offer different avenues for studying CD48 function, although antibodies remain essential for validation.
- Supply Chain Disruptions: Global supply chain disruptions, as witnessed in recent years, can impact the availability of raw materials and the timely delivery of antibodies, posing a challenge for consistent research workflows.
- Market Saturation in Certain Segments: While the overall market is growing, some established antibody applications might experience increased competition and price pressures, requiring continuous innovation to maintain market share. The impact of these challenges could lead to a 5% slowdown in projected growth if not adequately addressed.
Emerging Opportunities in CD48 Antibody
The CD48 antibody market is ripe with emerging opportunities:
- Therapeutic Applications: The growing understanding of CD48's role in immune regulation and disease pathogenesis presents significant opportunities for developing therapeutic antibodies targeting CD48 for autoimmune diseases, inflammatory conditions, and even certain cancers.
- Personalized Medicine and Biomarker Development: CD48 can serve as a valuable biomarker for disease progression and treatment response. Opportunities exist in developing CD48 antibody-based assays for personalized medicine approaches.
- Combinatorial Therapies: Investigating the synergistic effects of CD48 antibodies in combination with other immunotherapies or targeted drugs holds immense potential for developing more effective treatment strategies.
- Point-of-Care Diagnostics: Development of rapid and sensitive CD48 antibody-based diagnostic tests for point-of-care applications could revolutionize disease detection and management.
- Emerging Markets: Expansion into underdeveloped and emerging markets with growing investments in healthcare and life sciences research presents a significant opportunity for market growth and penetration.
Leading Players in the CD48 Antibody Market
- Sino Biological, Inc.
- Cell Signaling Technology, Inc.
- Bio-Techne
- OriGene Technologies, Inc.
- LifeSpan BioSciences, Inc
- Abbexa
- MyBiosource, Inc.
- Biorbyt
- Creative Biolabs
- GeneTex
- Arigo Biolaboratories Corp.
- Abcam
- RayBiotech, Inc.
- Assay Genie
Key Developments in CD48 Antibody Industry
- 2023/2024: Increased research focus on CD48's role in CAR-T cell therapy efficacy and resistance mechanisms.
- 2023/2024: Launch of novel, highly validated CD48 antibodies with enhanced performance for multiplex immunofluorescence applications by several key players.
- 2022/2023: Significant advancements in recombinant antibody production leading to improved batch consistency and reduced lot-to-lot variability.
- 2021/2022: Growing interest in CD48 as a target for immunomodulatory drugs in the context of autoimmune diseases and inflammatory disorders.
- 2020/2021: Expansion of CD48 antibody applications in single-cell sequencing workflows for immune profiling.
- 2019/2020: Increased collaborations between antibody manufacturers and pharmaceutical companies for the development of therapeutic CD48 antibodies.
Future Outlook for CD48 Antibody Market
The future outlook for the CD48 antibody market is exceptionally bright, driven by continuous scientific innovation and an expanding understanding of immune system biology. The increasing demand for precise diagnostic tools and targeted immunotherapies will continue to fuel market growth. Opportunities in therapeutic antibody development, personalized medicine, and the exploration of combinatorial treatment strategies present significant avenues for expansion. As research delves deeper into the intricate roles of CD48 in various physiological and pathological processes, the demand for high-quality, specialized CD48 antibodies is projected to soar, solidifying its position as a critical reagent in the life sciences. The market is expected to witness a steady CAGR of over 12%, reaching over $50 billion by 2033.
cd48 antibody Segmentation
-
1. Application
- 1.1. Flow Cytometry
- 1.2. ELISA
- 1.3. Western Blot
- 1.4. Immunoprecipitation
- 1.5. Immunofluorescence
- 1.6. Others
-
2. Types
- 2.1. Monoclonal Antibody
- 2.2. Polyclonal Antibody
cd48 antibody Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
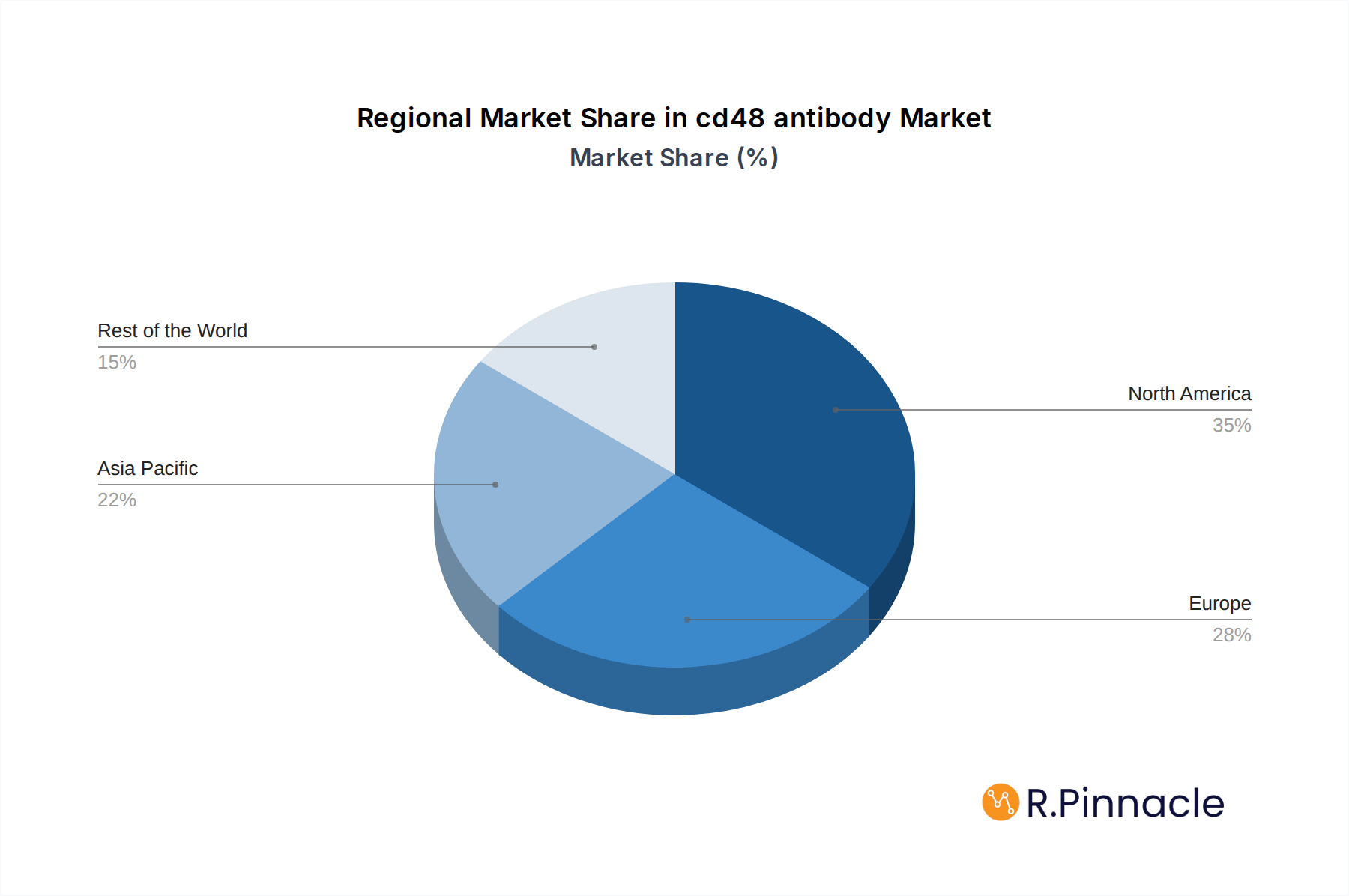
cd48 antibody Regional Market Share

Geographic Coverage of cd48 antibody
cd48 antibody REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Flow Cytometry
- 5.1.2. ELISA
- 5.1.3. Western Blot
- 5.1.4. Immunoprecipitation
- 5.1.5. Immunofluorescence
- 5.1.6. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Monoclonal Antibody
- 5.2.2. Polyclonal Antibody
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Flow Cytometry
- 6.1.2. ELISA
- 6.1.3. Western Blot
- 6.1.4. Immunoprecipitation
- 6.1.5. Immunofluorescence
- 6.1.6. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Monoclonal Antibody
- 6.2.2. Polyclonal Antibody
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Flow Cytometry
- 7.1.2. ELISA
- 7.1.3. Western Blot
- 7.1.4. Immunoprecipitation
- 7.1.5. Immunofluorescence
- 7.1.6. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Monoclonal Antibody
- 7.2.2. Polyclonal Antibody
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Flow Cytometry
- 8.1.2. ELISA
- 8.1.3. Western Blot
- 8.1.4. Immunoprecipitation
- 8.1.5. Immunofluorescence
- 8.1.6. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Monoclonal Antibody
- 8.2.2. Polyclonal Antibody
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Flow Cytometry
- 9.1.2. ELISA
- 9.1.3. Western Blot
- 9.1.4. Immunoprecipitation
- 9.1.5. Immunofluorescence
- 9.1.6. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Monoclonal Antibody
- 9.2.2. Polyclonal Antibody
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific cd48 antibody Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Flow Cytometry
- 10.1.2. ELISA
- 10.1.3. Western Blot
- 10.1.4. Immunoprecipitation
- 10.1.5. Immunofluorescence
- 10.1.6. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Monoclonal Antibody
- 10.2.2. Polyclonal Antibody
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Sino Biological
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Inc.
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Cell Signaling Technology
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Inc.
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Bio-Techne
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 OriGene Technologies
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Inc.
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 LifeSpan BioSciences
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Inc
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Abbexa
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 MyBiosource
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Inc.
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Biorbyt
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Creative Biolabs
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 GeneTex
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Arigo Biolaboratories Corp.
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Abcam
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 RayBiotech
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Inc.
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Assay Genie
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.1 Sino Biological
List of Figures
- Figure 1: Global cd48 antibody Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global cd48 antibody Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America cd48 antibody Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America cd48 antibody Volume (K), by Application 2025 & 2033
- Figure 5: North America cd48 antibody Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America cd48 antibody Volume Share (%), by Application 2025 & 2033
- Figure 7: North America cd48 antibody Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America cd48 antibody Volume (K), by Types 2025 & 2033
- Figure 9: North America cd48 antibody Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America cd48 antibody Volume Share (%), by Types 2025 & 2033
- Figure 11: North America cd48 antibody Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America cd48 antibody Volume (K), by Country 2025 & 2033
- Figure 13: North America cd48 antibody Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America cd48 antibody Volume Share (%), by Country 2025 & 2033
- Figure 15: South America cd48 antibody Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America cd48 antibody Volume (K), by Application 2025 & 2033
- Figure 17: South America cd48 antibody Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America cd48 antibody Volume Share (%), by Application 2025 & 2033
- Figure 19: South America cd48 antibody Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America cd48 antibody Volume (K), by Types 2025 & 2033
- Figure 21: South America cd48 antibody Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America cd48 antibody Volume Share (%), by Types 2025 & 2033
- Figure 23: South America cd48 antibody Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America cd48 antibody Volume (K), by Country 2025 & 2033
- Figure 25: South America cd48 antibody Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America cd48 antibody Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe cd48 antibody Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe cd48 antibody Volume (K), by Application 2025 & 2033
- Figure 29: Europe cd48 antibody Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe cd48 antibody Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe cd48 antibody Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe cd48 antibody Volume (K), by Types 2025 & 2033
- Figure 33: Europe cd48 antibody Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe cd48 antibody Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe cd48 antibody Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe cd48 antibody Volume (K), by Country 2025 & 2033
- Figure 37: Europe cd48 antibody Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe cd48 antibody Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa cd48 antibody Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa cd48 antibody Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa cd48 antibody Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa cd48 antibody Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa cd48 antibody Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa cd48 antibody Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa cd48 antibody Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa cd48 antibody Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa cd48 antibody Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa cd48 antibody Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa cd48 antibody Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa cd48 antibody Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific cd48 antibody Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific cd48 antibody Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific cd48 antibody Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific cd48 antibody Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific cd48 antibody Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific cd48 antibody Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific cd48 antibody Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific cd48 antibody Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific cd48 antibody Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific cd48 antibody Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific cd48 antibody Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific cd48 antibody Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 3: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 5: Global cd48 antibody Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global cd48 antibody Volume K Forecast, by Region 2020 & 2033
- Table 7: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 9: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 11: Global cd48 antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global cd48 antibody Volume K Forecast, by Country 2020 & 2033
- Table 13: United States cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 21: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 23: Global cd48 antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global cd48 antibody Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 33: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 35: Global cd48 antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global cd48 antibody Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 57: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 59: Global cd48 antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global cd48 antibody Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global cd48 antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global cd48 antibody Volume K Forecast, by Application 2020 & 2033
- Table 75: Global cd48 antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global cd48 antibody Volume K Forecast, by Types 2020 & 2033
- Table 77: Global cd48 antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global cd48 antibody Volume K Forecast, by Country 2020 & 2033
- Table 79: China cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific cd48 antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific cd48 antibody Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the cd48 antibody?
The projected CAGR is approximately 6.8%.
2. Which companies are prominent players in the cd48 antibody?
Key companies in the market include Sino Biological, Inc., Cell Signaling Technology, Inc., Bio-Techne, OriGene Technologies, Inc., LifeSpan BioSciences, Inc, Abbexa, MyBiosource, Inc., Biorbyt, Creative Biolabs, GeneTex, Arigo Biolaboratories Corp., Abcam, RayBiotech, Inc., Assay Genie.
3. What are the main segments of the cd48 antibody?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4350.00, USD 6525.00, and USD 8700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "cd48 antibody," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the cd48 antibody report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the cd48 antibody?
To stay informed about further developments, trends, and reports in the cd48 antibody, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
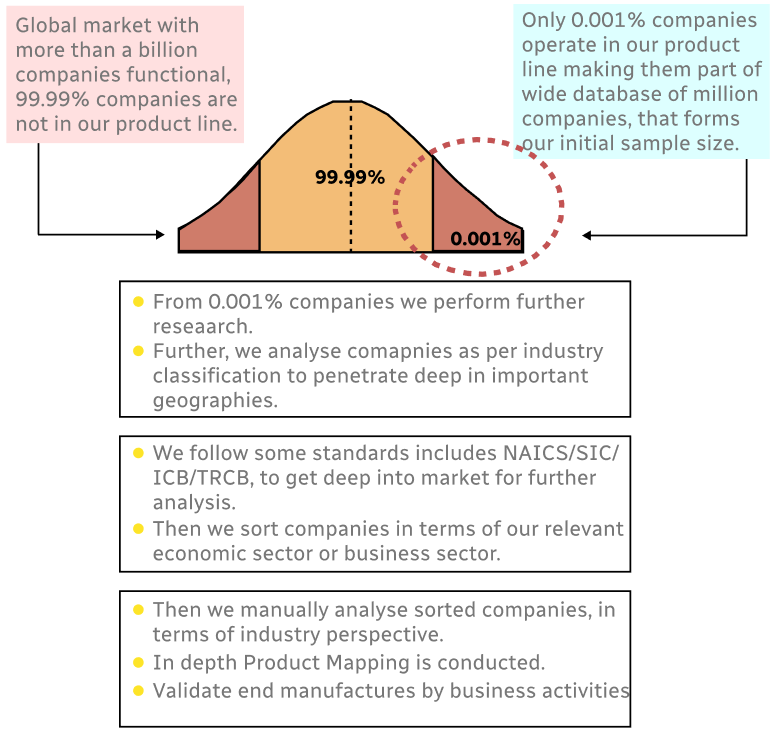
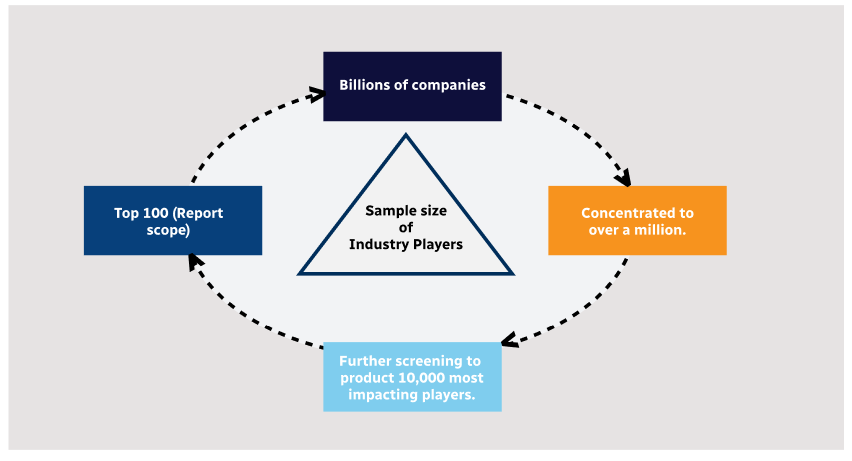
Step 1 - Identification of Relevant Samples Size from Population Database
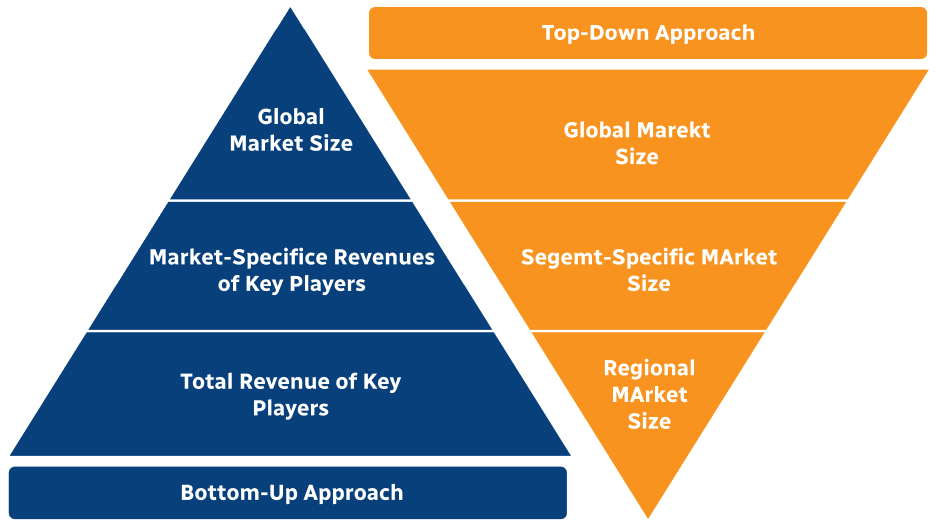
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
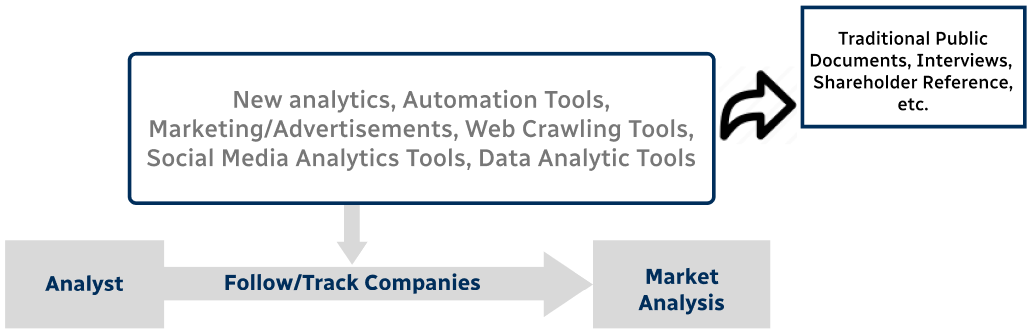
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


