Key Insights
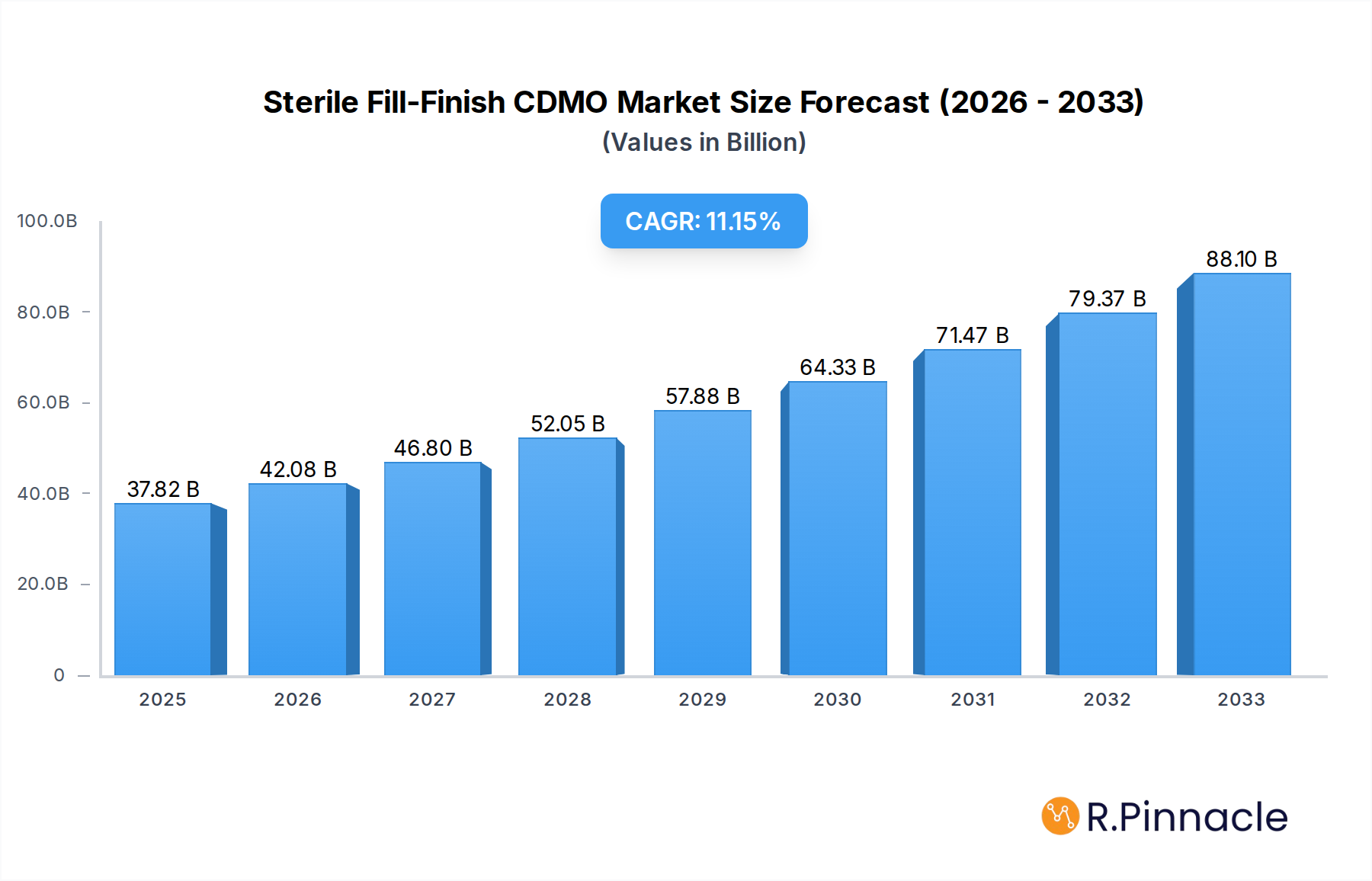
The global Sterile Fill-Finish Contract Development and Manufacturing Organization (CDMO) market is poised for substantial growth, reaching an estimated $37.82 billion in 2025. This expansion is driven by an escalating demand for biopharmaceuticals, vaccines, and biosimilars, coupled with an increasing trend among pharmaceutical companies to outsource complex manufacturing processes. The market is projected to witness a robust Compound Annual Growth Rate (CAGR) of 11.2% over the forecast period of 2025-2033. Key factors fueling this growth include the rising prevalence of chronic diseases, the burgeoning pipeline of biologics, and the ongoing innovation in drug delivery systems like prefilled syringes and cartridges. Furthermore, the growing complexity of drug formulations and the need for specialized sterile environments necessitate the expertise offered by CDMOs, making them indispensable partners for drug developers. The increasing focus on drug product lifecycle management and the desire for cost-efficiency further bolster the reliance on specialized CDMO services.

Sterile Fill-Finish CDMO Market Size (In Billion)

Geographically, North America and Europe currently dominate the market, attributed to the presence of major pharmaceutical players and advanced healthcare infrastructure. However, the Asia Pacific region is anticipated to emerge as a significant growth engine due to expanding pharmaceutical manufacturing capabilities, increasing R&D investments, and a growing domestic demand for advanced therapeutics. The market is segmented by application, with Vaccines, Biologics and Biosimilar, and Generics representing the primary demand areas. Prefilled syringes and vials are expected to be the leading types of fill-finish solutions, reflecting their widespread use in various therapeutic areas. Key players in the market are investing heavily in expanding their capacities, adopting advanced technologies such as isolator technology and automated filling lines, and focusing on strategic collaborations and mergers to gain a competitive edge and cater to the evolving needs of the pharmaceutical industry.
Sterile Fill-Finish CDMO Company Market Share

Here is your SEO-optimized, reader-centric report description for the Sterile Fill-Finish CDMO market:
Sterile Fill-Finish CDMO Market Structure & Innovation Trends
This comprehensive report delves into the intricate structure of the sterile fill-finish CDMO market, a critical segment within the pharmaceutical outsourcing landscape valued at billions. We analyze market concentration, identifying key players and their respective shares, estimated to be in the hundreds of billions. Innovation drivers, such as advancements in aseptic processing technologies and specialized filling solutions, are meticulously examined. The report scrutinizes the evolving regulatory frameworks, including GMP compliance and emerging guidelines that shape operational standards. We also assess the threat of product substitutes and the influence of end-user demographics, particularly the rising demand for biologics and complex drug formulations, on market dynamics. Furthermore, extensive analysis of Mergers & Acquisitions (M&A) activities, with deal values reaching billions, provides insights into strategic consolidations and market expansion.
- Market Concentration: Analysis of market share distribution among leading CDMOs.
- Innovation Drivers: Focus on technological advancements and R&D investments.
- Regulatory Frameworks: Examination of global and regional compliance standards.
- Product Substitutes: Assessment of alternative manufacturing and delivery methods.
- End-User Demographics: Impact of patient populations and disease trends.
- M&A Activities: Detailed review of strategic partnerships and acquisitions.
Sterile Fill-Finish CDMO Market Dynamics & Trends
The sterile fill-finish contract development and manufacturing organization (CDMO) market is experiencing robust growth, driven by an increasing demand for outsourced pharmaceutical manufacturing solutions. The global market size is projected to exceed billions by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 10-15% during the forecast period (2025-2033). This sustained expansion is fueled by several key factors. Firstly, the burgeoning biopharmaceutical sector, with its pipeline of complex biologics, biosimilars, and novel therapies requiring specialized sterile fill-finish capabilities, is a primary growth accelerant. Companies are increasingly relying on CDMOs to navigate the complexities of aseptic processing, lyophilization, and parenteral drug product manufacturing. Secondly, technological disruptions are reshaping the market. Advancements in automation, single-use technologies, and advanced sterile filtration systems are enhancing efficiency, reducing contamination risks, and improving overall product quality. The adoption of isolator technology and rapid sterilization techniques are becoming industry standards. Thirdly, evolving consumer preferences and healthcare trends are contributing significantly. The growing prevalence of chronic diseases and the increasing demand for injectable drug delivery systems, particularly prefilled syringes and vials for vaccines and biologics, are driving the need for specialized fill-finish services. The market penetration of advanced therapeutic modalities like cell and gene therapies further amplifies this demand. Finally, the competitive dynamics are characterized by both intense rivalry among established players and the emergence of new, specialized CDMOs. Strategic collaborations, capacity expansions, and the pursuit of specialized niche capabilities are key strategies employed by companies to gain a competitive edge. The outsourcing trend is expected to continue as pharmaceutical companies focus on core competencies like R&D and drug discovery, while entrusting manufacturing to expert partners. The market penetration of outsourced sterile fill-finish services is projected to rise, driven by cost-effectiveness, access to specialized expertise, and the ability to scale operations rapidly.
Dominant Regions & Segments in Sterile Fill-Finish CDMO
North America currently leads the sterile fill-finish CDMO market, with an estimated market share of billions, driven by its robust pharmaceutical industry, significant investments in biopharmaceutical research and development, and a high concentration of major pharmaceutical and biotechnology companies. The United States, in particular, is a powerhouse, benefiting from supportive government policies, a well-established healthcare infrastructure, and a strong ecosystem of innovation. Economic policies promoting R&D and tax incentives for manufacturing further bolster its dominance.
Key Dominance Drivers in North America:
- Extensive R&D Pipeline: A high volume of novel biologics and vaccines in development.
- Advanced Manufacturing Infrastructure: State-of-the-art facilities and cutting-edge technologies.
- Skilled Workforce: Availability of highly trained professionals in sterile manufacturing.
- Favorable Regulatory Environment: Streamlined approval processes and strong regulatory oversight.
The Biologics and Biosimilar segment within the Application category represents another area of significant dominance, accounting for billions in market value. The escalating demand for biologics, driven by their efficacy in treating complex diseases such as cancer, autoimmune disorders, and rare genetic conditions, underpins this segment's strong performance. Biosimilars, as cost-effective alternatives to established biologic drugs, are also contributing to market expansion, requiring specialized sterile fill-finish capabilities for their production.
Key Dominance Drivers in the Biologics and Biosimilar Segment:
- Increasing Prevalence of Chronic Diseases: Growing patient populations requiring advanced therapeutic interventions.
- Pipeline Expansion: A surge in the development of new biologic entities and biosimilars.
- Therapeutic Efficacy: Superior treatment outcomes offered by biologic drugs.
- Cost-Containment Pressures: Demand for affordable biosimilar alternatives.
Among the Types, Prefilled Syringes are witnessing substantial growth and are a key driver of market value, estimated in billions. The convenience, accuracy, and patient compliance offered by prefilled syringes make them a preferred choice for a wide range of injectable medications, particularly for self-administration. This trend is further amplified by the global vaccination initiatives and the increasing use of complex biologics delivered via parenteral routes.
Key Dominance Drivers in the Prefilled Syringes Segment:
- Patient Convenience and Safety: Enhanced ease of use and reduced risk of medication errors.
- Growing Demand for Biologics: Many advanced therapies are formulated in prefilled syringes.
- Self-Administration Trends: Growing patient preference for at-home treatment options.
- Technological Advancements: Innovations in syringe design and functionality.
Sterile Fill-Finish CDMO Product Innovations
Sterile fill-finish CDMOs are at the forefront of innovation, developing advanced solutions to meet the evolving demands of the pharmaceutical industry. Recent product developments focus on enhancing aseptic processing capabilities, including sophisticated isolator technologies and integrated robotic systems that minimize human intervention and contamination risks. The market is seeing an increased offering of specialized filling for high-potency active pharmaceutical ingredients (HPAPIs) and biologics, often requiring lyophilization or aseptic crystallization. These advancements provide competitive advantages by ensuring product integrity, extending shelf-life, and enabling the delivery of complex therapeutic agents. The focus on single-use technologies and sterile powder filling further exemplifies the industry's commitment to efficiency and flexibility, catering to a diverse range of drug formulations.
Report Scope & Segmentation Analysis
This report provides an in-depth analysis of the sterile fill-finish CDMO market, segmented across key application areas and product types. The Application segment encompasses Vaccines, Biologics and Biosimilar, Generics, and Other therapeutic categories. The Biologics and Biosimilar segment, valued in billions, is projected to exhibit significant growth due to the expanding pipeline and market penetration of these advanced therapies. The Types segmentation includes Vials, Prefilled Syringes, Cartridges, and Others. The Prefilled Syringes segment, also valued in billions, is anticipated to witness robust growth driven by patient convenience and the increasing demand for self-administered medications. Competitive dynamics within each segment are analyzed, with projections on market size and growth rates, reflecting the strategic importance of each area to CDMOs.
Key Drivers of Sterile Fill-Finish CDMO Growth
The sterile fill-finish CDMO market's impressive growth, valued in billions, is propelled by a confluence of critical factors.
- Technological Advancements: The continuous development and adoption of advanced aseptic processing technologies, including isolators, robotic filling systems, and advanced filtration, significantly improve efficiency and reduce contamination risks.
- Expanding Biologics Pipeline: The burgeoning number of biologic drugs, biosimilars, and novel therapeutic modalities requiring specialized sterile manufacturing expertise fuels outsourcing demand.
- Cost-Effectiveness and Specialization: Pharmaceutical companies increasingly outsource to leverage the specialized expertise, economies of scale, and reduced capital investment offered by CDMOs.
- Regulatory Compliance: Stringent regulatory requirements for sterile manufacturing drive the need for specialized CDMOs equipped to meet global standards.
Challenges in the Sterile Fill-Finish CDMO Sector
Despite its strong growth trajectory, the sterile fill-finish CDMO sector, with a market size in the billions, faces several significant challenges.
- Regulatory Hurdles and Evolving Standards: Navigating complex and ever-changing global regulatory landscapes, including stringent GMP compliance and inspection readiness, can be resource-intensive.
- Supply Chain Volatility: Disruptions in the supply of critical raw materials, components, and specialized equipment can impact production timelines and costs.
- Intense Competition and Pricing Pressures: The presence of numerous established and emerging CDMOs leads to fierce competition, often resulting in pricing pressures that can affect profit margins.
- Talent Shortage: A scarcity of highly skilled personnel with expertise in aseptic processing, quality control, and regulatory affairs poses a continuous challenge.
Emerging Opportunities in Sterile Fill-Finish CDMO
The sterile fill-finish CDMO market, projected to reach billions, is ripe with emerging opportunities.
- Expansion into Advanced Therapies: The growing field of cell and gene therapies, requiring specialized sterile handling and formulation, presents a significant opportunity for CDMOs with relevant expertise.
- Geographic Expansion: Tapping into emerging markets in Asia-Pacific and Latin America, where pharmaceutical manufacturing capabilities are developing rapidly, offers substantial growth potential.
- Technological Integration: Embracing digital transformation, including AI-driven process optimization and advanced analytics, can enhance efficiency and predictive maintenance.
- Specialized Niche Services: Developing expertise in niche areas like high-potency drug filling, sterile powders, and complex parenteral formulations can create distinct competitive advantages.
Leading Players in the Sterile Fill-Finish CDMO Market
Baxter BioPharma Solutions Boehringer Ingelheim Vetter Pharma Fresenius Kabi Pfizer CentreOne Aenova WuXi Biologics Jubilant HollisterStier Bushu Pharmaceuticals LSNE Contract Manufacturing Ajinomoto Bio-Pharma Services CMIC CMO GRAM (Grand River Aseptic Manufacturing) TAIYO Pharma Tech Co.,Ltd. HALIX Cognate BioServices Afton Scientific Novasep Emergent BioSolutions Seikagaku Jiangshu YAOHAI Bio-pharmaceutical Akron Biotech Symbiosis Pharmaceutical Services Techdow Vigene Biosciences
Key Developments in Sterile Fill-Finish CDMO Industry
- 2023: Several CDMOs announced significant capacity expansions for biologics and vaccine fill-finish operations, responding to increased global demand.
- 2022: Increased investment in advanced isolator technology and automation to enhance aseptic capabilities and reduce human intervention.
- 2021: Growing trend of CDMOs specializing in fill-finish for novel therapeutic modalities like mRNA vaccines and cell therapies.
- 2020: Strategic mergers and acquisitions aimed at consolidating market share and expanding service portfolios in the sterile fill-finish space.
- 2019: Enhanced focus on single-use technologies to improve flexibility and reduce cross-contamination risks in sterile fill-finish processes.
Future Outlook for Sterile Fill-Finish CDMO Market
The future outlook for the sterile fill-finish CDMO market, valued in billions, is exceptionally positive, driven by sustained demand for outsourced sterile manufacturing. The continued expansion of the biologics pipeline, coupled with the rise of personalized medicine and advanced therapies, will necessitate specialized fill-finish expertise. Pharmaceutical companies will increasingly rely on CDMOs to manage the complexities of aseptic processing, ensuring product quality and regulatory compliance. Strategic investments in new technologies, capacity expansion, and specialized capabilities will be crucial for CDMOs to maintain a competitive edge and capitalize on emerging opportunities in a dynamic global market.
Sterile Fill-Finish CDMO Segmentation
-
1. Application
- 1.1. Vaccines
- 1.2. Biologics and Biosimilar
- 1.3. Generics
- 1.4. Other
-
2. Types
- 2.1. Vials
- 2.2. Prefilled Syringes
- 2.3. Cartridges
- 2.4. Others
Sterile Fill-Finish CDMO Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
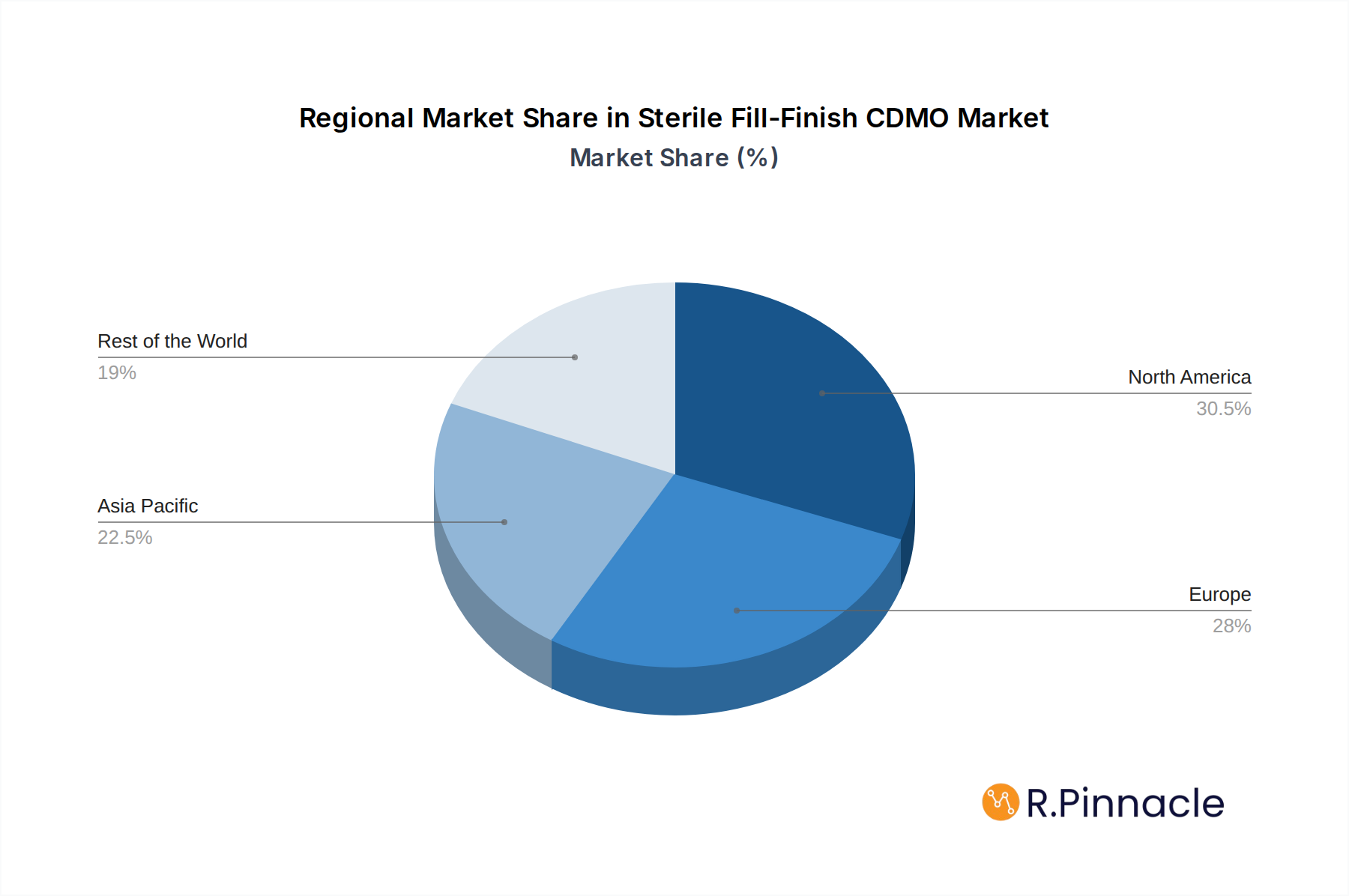
Sterile Fill-Finish CDMO Regional Market Share

Geographic Coverage of Sterile Fill-Finish CDMO
Sterile Fill-Finish CDMO REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.2% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Vaccines
- 5.1.2. Biologics and Biosimilar
- 5.1.3. Generics
- 5.1.4. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Vials
- 5.2.2. Prefilled Syringes
- 5.2.3. Cartridges
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Vaccines
- 6.1.2. Biologics and Biosimilar
- 6.1.3. Generics
- 6.1.4. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Vials
- 6.2.2. Prefilled Syringes
- 6.2.3. Cartridges
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Vaccines
- 7.1.2. Biologics and Biosimilar
- 7.1.3. Generics
- 7.1.4. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Vials
- 7.2.2. Prefilled Syringes
- 7.2.3. Cartridges
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Vaccines
- 8.1.2. Biologics and Biosimilar
- 8.1.3. Generics
- 8.1.4. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Vials
- 8.2.2. Prefilled Syringes
- 8.2.3. Cartridges
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Vaccines
- 9.1.2. Biologics and Biosimilar
- 9.1.3. Generics
- 9.1.4. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Vials
- 9.2.2. Prefilled Syringes
- 9.2.3. Cartridges
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Sterile Fill-Finish CDMO Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Vaccines
- 10.1.2. Biologics and Biosimilar
- 10.1.3. Generics
- 10.1.4. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Vials
- 10.2.2. Prefilled Syringes
- 10.2.3. Cartridges
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Baxter BioPharma Solutions
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Boehringer Ingelheim
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Vetter Pharma
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Fresenius Kabi
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Pfizer CentreOne
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Aenova
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 WuXi Biologics
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Jubilant HollisterStier
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Bushu Pharmaceuticals
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 LSNE Contract Manufacturing
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Ajinomoto Bio-Pharma Services
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 CMIC CMO
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 GRAM (Grand River Aseptic Manufacturing)
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 TAIYO Pharma Tech Co.Ltd.
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 HALIX
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Cognate BioServices
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Afton Scientific
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Novasep
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Emergent BioSolutions
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Seikagaku
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.21 Jiangshu YAOHAI Bio-pharmaceutical
- 11.2.21.1. Overview
- 11.2.21.2. Products
- 11.2.21.3. SWOT Analysis
- 11.2.21.4. Recent Developments
- 11.2.21.5. Financials (Based on Availability)
- 11.2.22 Akron Biotech
- 11.2.22.1. Overview
- 11.2.22.2. Products
- 11.2.22.3. SWOT Analysis
- 11.2.22.4. Recent Developments
- 11.2.22.5. Financials (Based on Availability)
- 11.2.23 Symbiosis Pharmaceutical Services
- 11.2.23.1. Overview
- 11.2.23.2. Products
- 11.2.23.3. SWOT Analysis
- 11.2.23.4. Recent Developments
- 11.2.23.5. Financials (Based on Availability)
- 11.2.24 Techdow
- 11.2.24.1. Overview
- 11.2.24.2. Products
- 11.2.24.3. SWOT Analysis
- 11.2.24.4. Recent Developments
- 11.2.24.5. Financials (Based on Availability)
- 11.2.25 Vigene Biosciences
- 11.2.25.1. Overview
- 11.2.25.2. Products
- 11.2.25.3. SWOT Analysis
- 11.2.25.4. Recent Developments
- 11.2.25.5. Financials (Based on Availability)
- 11.2.1 Baxter BioPharma Solutions
List of Figures
- Figure 1: Global Sterile Fill-Finish CDMO Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Sterile Fill-Finish CDMO Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Sterile Fill-Finish CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Sterile Fill-Finish CDMO Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Sterile Fill-Finish CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Sterile Fill-Finish CDMO Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Sterile Fill-Finish CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Sterile Fill-Finish CDMO Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Sterile Fill-Finish CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Sterile Fill-Finish CDMO Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Sterile Fill-Finish CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Sterile Fill-Finish CDMO Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Sterile Fill-Finish CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Sterile Fill-Finish CDMO Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Sterile Fill-Finish CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Sterile Fill-Finish CDMO Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Sterile Fill-Finish CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Sterile Fill-Finish CDMO Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Sterile Fill-Finish CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Sterile Fill-Finish CDMO Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Sterile Fill-Finish CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Sterile Fill-Finish CDMO Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Sterile Fill-Finish CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Sterile Fill-Finish CDMO Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Sterile Fill-Finish CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Sterile Fill-Finish CDMO Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Sterile Fill-Finish CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Sterile Fill-Finish CDMO Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Sterile Fill-Finish CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Sterile Fill-Finish CDMO Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Sterile Fill-Finish CDMO Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Sterile Fill-Finish CDMO Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Sterile Fill-Finish CDMO Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Sterile Fill-Finish CDMO?
The projected CAGR is approximately 11.2%.
2. Which companies are prominent players in the Sterile Fill-Finish CDMO?
Key companies in the market include Baxter BioPharma Solutions, Boehringer Ingelheim, Vetter Pharma, Fresenius Kabi, Pfizer CentreOne, Aenova, WuXi Biologics, Jubilant HollisterStier, Bushu Pharmaceuticals, LSNE Contract Manufacturing, Ajinomoto Bio-Pharma Services, CMIC CMO, GRAM (Grand River Aseptic Manufacturing), TAIYO Pharma Tech Co.,Ltd., HALIX, Cognate BioServices, Afton Scientific, Novasep, Emergent BioSolutions, Seikagaku, Jiangshu YAOHAI Bio-pharmaceutical, Akron Biotech, Symbiosis Pharmaceutical Services, Techdow, Vigene Biosciences.
3. What are the main segments of the Sterile Fill-Finish CDMO?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Sterile Fill-Finish CDMO," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Sterile Fill-Finish CDMO report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Sterile Fill-Finish CDMO?
To stay informed about further developments, trends, and reports in the Sterile Fill-Finish CDMO, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
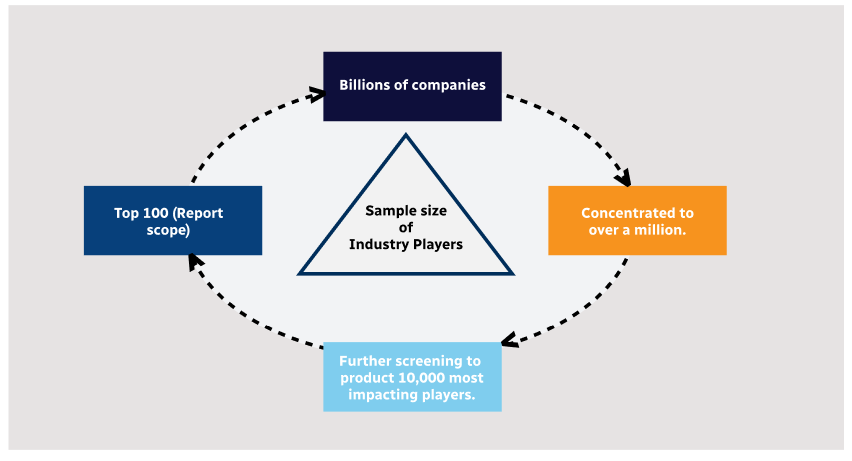
Step 1 - Identification of Relevant Samples Size from Population Database
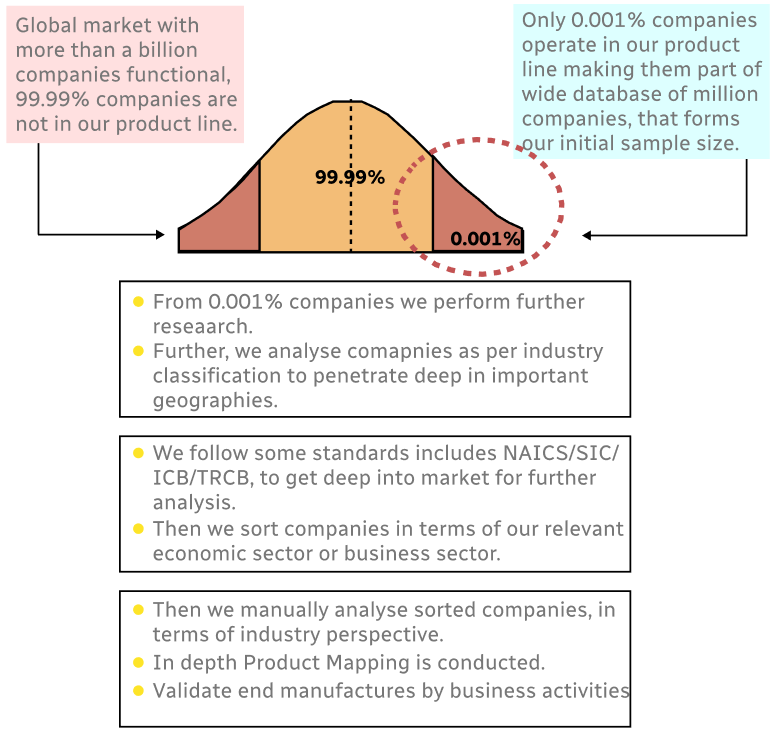
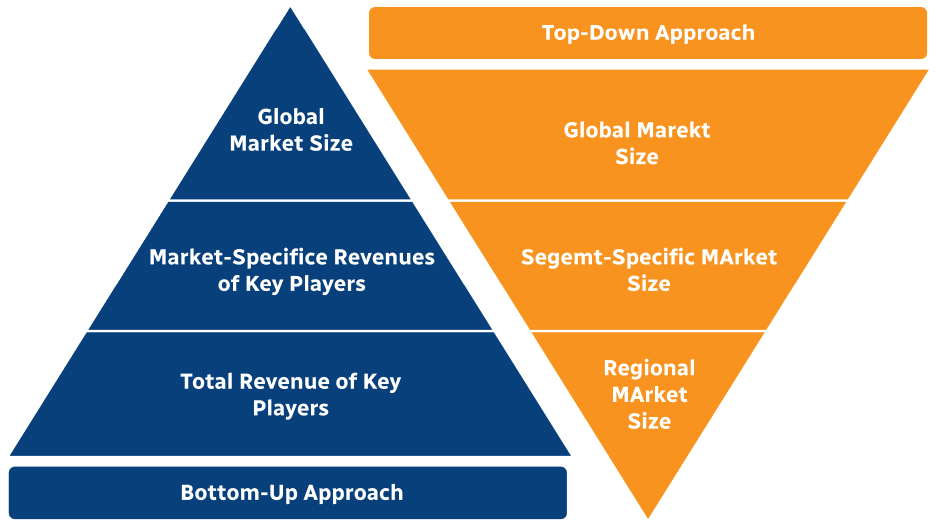
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


