Key Insights
The global synthetic peptide vaccine market is poised for significant expansion, with an estimated market size of $7.77 billion in 2025. This growth is fueled by a projected Compound Annual Growth Rate (CAGR) of 10.81% throughout the forecast period of 2025-2033. The increasing prevalence of chronic diseases, particularly various forms of cancer such as prostate, breast, gastric, and lung cancer, is a primary driver for this market. Synthetic peptide vaccines offer a targeted and potentially safer alternative to traditional therapies, attracting substantial investment in research and development. Advances in synthesis technologies, including Solution Phase Peptide Synthesis (SPS) and Solid Phase Peptide Synthesis (SPPS), are enhancing the efficiency and scalability of peptide production, making these vaccines more accessible and cost-effective. The growing understanding of the immune system and the development of personalized medicine approaches further bolster the demand for custom-designed synthetic peptide vaccines.
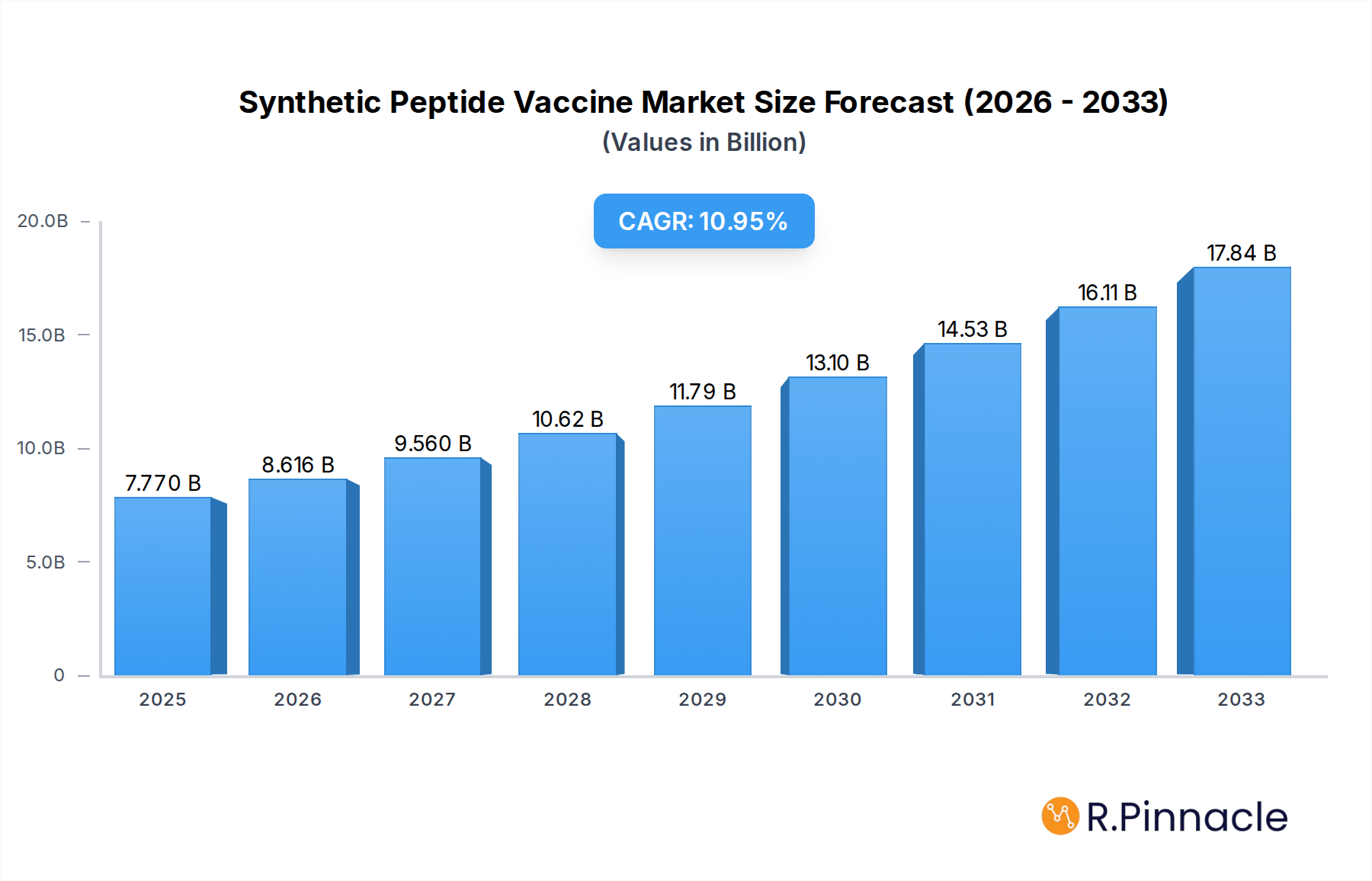
Synthetic Peptide Vaccine Market Size (In Billion)

Key trends shaping the synthetic peptide vaccine market include the rise of personalized oncology, where vaccines are tailored to individual patient tumor antigens, and the exploration of peptide vaccines for infectious diseases beyond cancer. The strategic initiatives undertaken by leading companies, such as mergers, acquisitions, and collaborative research efforts, are accelerating product development and market penetration. Furthermore, supportive government initiatives and increasing healthcare expenditure globally are contributing to market growth. While the market exhibits robust growth, challenges such as the high cost of development and manufacturing, stringent regulatory hurdles, and the need for extensive clinical validation for efficacy and safety may present some restraints. However, ongoing technological advancements and a deeper understanding of immunotherapies are expected to overcome these obstacles, solidifying the synthetic peptide vaccine market's upward trajectory.
Synthetic Peptide Vaccine Company Market Share

This in-depth market research report provides a definitive analysis of the global synthetic peptide vaccine market, covering historical data from 2019 to 2024, a base year of 2025, and a comprehensive forecast period extending to 2033. Leveraging high-ranking keywords for enhanced search visibility, this report is designed for industry professionals seeking actionable insights into market structure, dynamics, innovation trends, regional dominance, product developments, growth drivers, challenges, emerging opportunities, leading players, and future outlook. Our analysis encompasses key applications such as Prostate Cancer, Breast Cancer, Gastric Cancer, Lung Cancer, Pancreatic Cancer, and Others, along with synthesis types including Solution Phase Synthesis (SPS) and Solid Phase Peptide Synthesis (SPPS). The projected market value for the forecast period is estimated to reach billions, driven by significant advancements in therapeutic peptide development and increasing adoption in oncology.
Synthetic Peptide Vaccine Market Structure & Innovation Trends
The synthetic peptide vaccine market exhibits a moderately consolidated structure, with key players investing heavily in research and development to drive innovation. The primary innovation drivers include the precise targeting capabilities of peptide antigens, reduced immunogenicity compared to traditional vaccines, and the potential for personalized medicine approaches. Regulatory frameworks are evolving to accommodate these novel therapeutic modalities, with an increasing emphasis on clinical trial efficacy and safety. Product substitutes, while present in the broader vaccine landscape, face limitations in their ability to elicit specific T-cell responses, a key advantage of synthetic peptide vaccines. End-user demographics are primarily healthcare providers, research institutions, and pharmaceutical companies actively involved in oncology and immunology. Merger and acquisition (M&A) activities are on an upward trajectory, with an estimated billion in deal values observed historically, signaling strategic consolidation and expansion by major corporations.
- Market Concentration: Moderate, with significant R&D investment by leading entities.
- Innovation Drivers: Precision targeting, reduced immunogenicity, personalized medicine.
- Regulatory Frameworks: Evolving to support novel therapeutic peptide development.
- Product Substitutes: Limited in their ability to elicit specific T-cell responses.
- End-User Demographics: Healthcare providers, research institutions, pharmaceutical companies.
- M&A Activities: Increasing, with historical deal values estimated in the billions.
Synthetic Peptide Vaccine Market Dynamics & Trends
The synthetic peptide vaccine market is poised for substantial growth, propelled by a confluence of escalating cancer incidences worldwide and a paradigm shift towards more targeted and personalized immunotherapies. Technological advancements in peptide synthesis, purification, and formulation are continuously improving the efficacy and scalability of these vaccines, making them increasingly attractive for clinical development. The growing understanding of tumor immunology and the intricate mechanisms of immune evasion by cancer cells is further fueling research into novel peptide antigen designs that can effectively stimulate robust anti-tumor immune responses. Consumer preferences are increasingly leaning towards therapies with fewer side effects and higher specificity, characteristics that synthetic peptide vaccines are well-positioned to deliver. The competitive landscape is intensifying, characterized by strategic partnerships, collaborations, and significant investments in clinical trials aimed at bringing innovative peptide-based cancer vaccines to market. The compound annual growth rate (CAGR) for the synthetic peptide vaccine market is projected to be a significant percentage, driven by increasing market penetration in key oncology indications and the expansion of therapeutic applications beyond primary targets. Market penetration for synthetic peptide vaccines is expected to rise considerably as clinical data matures and regulatory approvals are secured for promising candidates.
Dominant Regions & Segments in Synthetic Peptide Vaccine
North America currently dominates the global synthetic peptide vaccine market, driven by robust healthcare infrastructure, substantial R&D investments, and a high prevalence of cancer diagnoses. The United States, in particular, plays a pivotal role due to its advanced pharmaceutical industry, leading research institutions, and supportive regulatory environment for novel therapeutics. Economic policies favoring innovation and significant government funding for cancer research further bolster its leading position.
Key Drivers for Regional Dominance:
- Economic Policies: Favorable funding for R&D and supportive intellectual property laws.
- Infrastructure: Advanced clinical trial networks and state-of-the-art manufacturing facilities.
- Regulatory Landscape: Streamlined approval processes for innovative oncology treatments.
- Prevalence of Cancer: High incidence rates of target cancers, creating a strong demand for effective therapies.
Within the application segments, Prostate Cancer and Breast Cancer represent the most significant markets for synthetic peptide vaccines, owing to their widespread occurrence and the ongoing efforts to develop more targeted and effective treatments. These segments benefit from extensive clinical research and a pipeline of peptide-based vaccine candidates.
Dominant Application Segments:
- Prostate Cancer: High unmet medical need and a significant focus on immunotherapy.
- Breast Cancer: Growing interest in personalized vaccines targeting specific tumor neoantigens.
In terms of synthesis types, Solid Phase Peptide Synthesis (SPPS) continues to be the predominant method due to its scalability, automation capabilities, and relatively lower cost for producing high-purity peptides required for vaccine development.
Dominant Synthesis Type:
- Solid Phase Peptide Synthesis (SPPS): Preferred for its efficiency, scalability, and cost-effectiveness in producing high-purity peptides for therapeutic applications.
The market is projected to witness continued growth in these dominant regions and segments, with increasing investments in clinical trials and the development of next-generation peptide vaccines.
Synthetic Peptide Vaccine Product Innovations
Recent product innovations in the synthetic peptide vaccine market are centered on enhancing immunogenicity, optimizing antigen design, and improving delivery mechanisms. These advancements aim to overcome the challenges of inducing potent and durable immune responses against cancer. Key innovations include the development of multi-epitope vaccines targeting a broader range of tumor-associated antigens, conjugation of peptides with immunomodulatory adjuvants, and the exploration of novel delivery platforms such as liposomes and nanoparticles. These efforts contribute to improved therapeutic efficacy and reduced side effects, positioning synthetic peptide vaccines as a competitive and promising class of cancer immunotherapies.
Report Scope & Segmentation Analysis
This report meticulously segments the synthetic peptide vaccine market by Application and Synthesis Type, providing granular insights into each category.
Application Segmentation:
- Prostate Cancer: This segment is driven by the substantial incidence of prostate cancer and the ongoing quest for improved treatment modalities, with projections indicating significant market growth.
- Breast Cancer: Characterized by a strong research focus on personalized oncology, this segment is expected to see considerable expansion driven by the development of tailored peptide vaccines.
- Gastric Cancer: While currently a smaller segment, gastric cancer applications are gaining traction with promising preclinical and early-stage clinical data.
- Lung Cancer: The high mortality rate and diverse subtypes of lung cancer present a significant opportunity for innovative peptide-based immunotherapies.
- Pancreatic Cancer: This notoriously difficult-to-treat cancer is a key focus area for new therapeutic approaches, including synthetic peptide vaccines.
- Others: This broad category encompasses emerging applications in various other cancer types and potentially other therapeutic areas, reflecting the versatility of peptide vaccine technology.
Synthesis Type Segmentation:
- Solution Phase Synthesis (SPS): While less common for large-scale vaccine production, SPS offers advantages for certain peptide structures and niche applications.
- Solid Phase Peptide Synthesis (SPPS): Remains the dominant methodology, offering scalability, automation, and cost-effectiveness crucial for the commercial production of therapeutic peptides.
Market sizes and growth projections for each segment are detailed within the report, reflecting the dynamic interplay of R&D activities, clinical trial outcomes, and market adoption.
Key Drivers of Synthetic Peptide Vaccine Growth
The synthetic peptide vaccine market is propelled by several key drivers that are reshaping the landscape of cancer immunotherapy. The increasing prevalence of various cancers globally necessitates the development of more effective and less toxic treatment options. Technological advancements in peptide synthesis and manufacturing are making these complex molecules more accessible and affordable. Furthermore, a deeper understanding of the immune system and tumor biology is enabling the design of highly specific and potent peptide antigens that can elicit targeted anti-tumor responses. The growing demand for personalized medicine, where treatments are tailored to an individual's genetic makeup and tumor characteristics, aligns perfectly with the capabilities of synthetic peptide vaccines to be designed around patient-specific neoantigens. Regulatory bodies are also showing increased support for innovative oncology therapies, streamlining approval pathways for promising candidates.
- Rising Cancer Incidence: Global surge in cancer cases fuels demand for novel therapies.
- Technological Advancements: Innovations in peptide synthesis and delivery systems.
- Immunological Understanding: Deeper insights into tumor immunity and T-cell activation.
- Personalized Medicine Trend: Tailoring treatments to individual patient profiles.
- Supportive Regulatory Environment: Evolving pathways for novel immunotherapies.
Challenges in the Synthetic Peptide Vaccine Sector
Despite the promising outlook, the synthetic peptide vaccine sector faces several challenges that could impede its growth. One of the primary hurdles is the inherent immunogenicity of peptides, which can sometimes lead to immune tolerance or inefficient immune responses. The complexity and cost associated with designing, synthesizing, and manufacturing highly specific peptide antigens for clinical use can be substantial. Furthermore, navigating the rigorous regulatory approval process for novel therapeutic vaccines requires extensive and lengthy clinical trials to demonstrate safety and efficacy, often exceeding billions in development costs. Ensuring consistent product quality and scalability for commercial production presents another significant challenge. Competitive pressures from other immunotherapy modalities, such as checkpoint inhibitors and CAR T-cell therapies, also necessitate continuous innovation and differentiation.
- Immunogenicity Challenges: Ensuring robust and effective immune responses.
- High Development Costs: Complex synthesis, manufacturing, and extensive clinical trials.
- Rigorous Regulatory Pathways: Long approval processes and stringent efficacy requirements.
- Scalability & Quality Control: Ensuring consistent large-scale production.
- Competitive Landscape: Pressure from established and emerging immunotherapy platforms.
Emerging Opportunities in Synthetic Peptide Vaccine
The synthetic peptide vaccine market is ripe with emerging opportunities driven by advancements in scientific understanding and technological innovation. The expansion of personalized oncology presents a significant avenue, with the development of patient-specific neoantigen vaccines becoming increasingly feasible. Exploring combination therapies, where synthetic peptide vaccines are used alongside other immunotherapies or conventional treatments, holds immense potential for synergistic effects and improved patient outcomes. The application of synthetic peptide vaccines beyond oncology, into infectious diseases and autoimmune disorders, represents another promising frontier. Furthermore, advancements in delivery technologies, such as mRNA-based delivery of peptide antigens or the use of novel adjuvants, could significantly enhance vaccine efficacy and patient compliance. The growing interest in prophylactic vaccines, particularly for high-risk populations or against prevalent infectious agents, also opens new avenues for synthetic peptide vaccine development.
- Personalized Neoantigen Vaccines: Tailoring treatments to individual tumor mutations.
- Combination Therapies: Synergistic effects with other immunotherapies and conventional treatments.
- Broader Therapeutic Applications: Expansion into infectious diseases and autoimmune disorders.
- Advanced Delivery Systems: Innovative platforms for enhanced efficacy and patient experience.
- Prophylactic Vaccine Development: Preventing diseases in high-risk populations.
Leading Players in the Synthetic Peptide Vaccine Market
- Anergis
- Antigen Express
- Apitope Technology
- Artificial Cell Technologies
- Benovus Bio
- Celgene Corporation
- Immatics biotechnologies
- Merck
- ImmunoCellular Therapeutics
- ISA Pharmaceuticals
- Lytix Biopharma
- Spotlight Innovation
- OncoTherapy Science
- Peptech
- Pharmasyntez
- Sumitomo Dainippon Pharma
- TapImmune
- Targovax
Key Developments in Synthetic Peptide Vaccine Industry
- 2023/10: Anergis announces successful completion of Phase 1 trial for its novel peptide vaccine candidate targeting HPV-related cancers, demonstrating promising safety and immunogenicity.
- 2023/08: Immatics biotechnologies announces positive interim data from its Phase 1b trial of a peptide-based therapeutic cancer vaccine, showing significant T-cell responses.
- 2023/05: Apitope Technology secures substantial funding to advance its platform for developing peptide-based therapies for autoimmune diseases, including promising early results.
- 2023/02: Merck and ISA Pharmaceuticals enter into a strategic collaboration to co-develop and commercialize a peptide-based immunotherapy for a specific type of solid tumor.
- 2022/11: Spotlight Innovation reports significant progress in its pipeline of synthetic peptide vaccines for glioblastoma, with ongoing preclinical studies showing encouraging efficacy.
- 2022/09: Targovax announces the initiation of a Phase 2 trial for its peptide-based vaccine targeting KRAS-mutated solid tumors, a significant step in advancing its pipeline.
- 2022/07: TapImmune announces expanded clinical trial for its lead peptide vaccine candidate in metastatic breast cancer, showing encouraging preliminary response rates.
- 2022/04: Sumitomo Dainippon Pharma acquires Peptech to bolster its oncology pipeline with peptide-based vaccine technologies.
- 2022/01: Lytix Biopharma receives regulatory approval to commence a Phase 1/2a clinical trial for its novel peptide-based cancer vaccine in combination with an immune checkpoint inhibitor.
- 2021/10: Antigen Express announces the successful development of a proprietary peptide design tool, accelerating the discovery of novel therapeutic peptide antigens.
- 2021/08: OncoTherapy Science advances its pipeline of personalized peptide vaccines for various solid tumors, with several candidates entering advanced preclinical development.
- 2021/05: Benovus Bio reports positive preclinical data for its peptide-based vaccine targeting pancreatic cancer, highlighting its potential to overcome tumor immunosuppression.
- 2021/02: Pharmasyntez announces a partnership to develop and manufacture synthetic peptide vaccines for the Russian market, expanding its global reach.
- 2020/11: Celgene Corporation (now part of Bristol Myers Squibb) continues to invest in its peptide-based immunotherapy pipeline, focusing on immune modulation and targeted delivery.
- 2020/08: Artificial Cell Technologies explores the potential of encapsulating synthetic peptides within artificial cells for improved stability and targeted delivery.
- 2020/05: ImmunoCellular Therapeutics advances its peptide vaccine programs, leveraging insights from earlier immune cell therapy trials.
Future Outlook for Synthetic Peptide Vaccine Market
The future outlook for the synthetic peptide vaccine market is exceptionally bright, with substantial growth accelerators anticipated over the forecast period. The increasing success of immunotherapies in oncology is paving the way for more sophisticated and targeted approaches, where synthetic peptide vaccines will play a pivotal role. Advancements in bioinformatics and artificial intelligence are expected to revolutionize peptide antigen design, enabling the rapid identification of highly immunogenic and tumor-specific epitopes. The growing clinical validation of these vaccines, coupled with the potential for combination therapies and personalized treatment strategies, will drive widespread adoption. Furthermore, ongoing research into new applications beyond oncology, such as in infectious diseases and autoimmune disorders, will diversify the market and unlock new revenue streams. The market is projected to reach billions in value, fueled by continuous innovation, strategic investments, and a growing pipeline of clinical candidates moving towards commercialization.
Synthetic Peptide Vaccine Segmentation
-
1. Application
- 1.1. Prostate Cancer
- 1.2. Breast Cancer
- 1.3. Gastric Cancer
- 1.4. Lung Cancer
- 1.5. Pancreatic Cancer
- 1.6. Others
-
2. Types
- 2.1. Solution Phase Synthesis (SPS)
- 2.2. Solid Phase Peptide Synthesis (SPPS)
Synthetic Peptide Vaccine Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
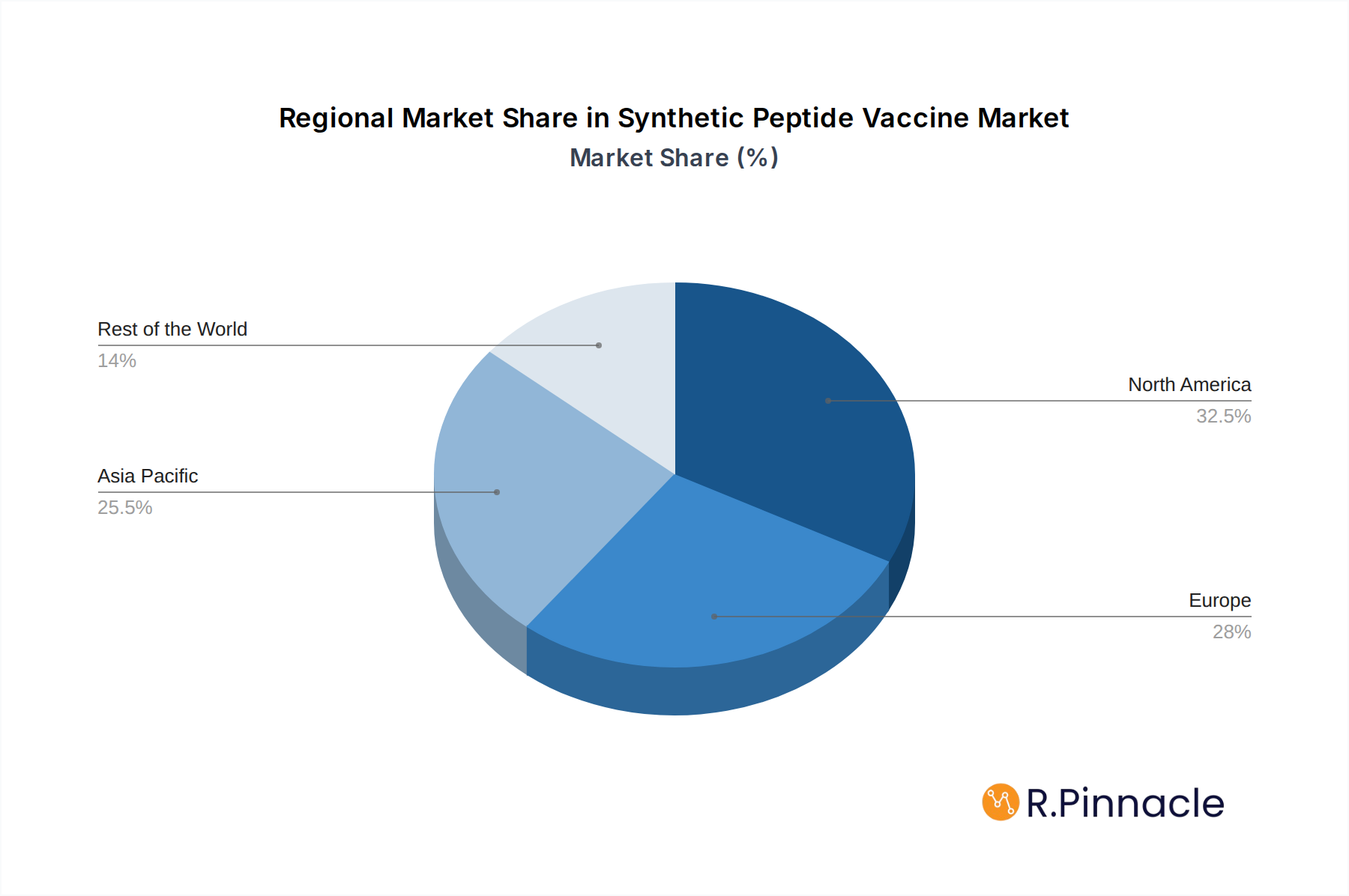
Synthetic Peptide Vaccine Regional Market Share

Geographic Coverage of Synthetic Peptide Vaccine
Synthetic Peptide Vaccine REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.81% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Prostate Cancer
- 5.1.2. Breast Cancer
- 5.1.3. Gastric Cancer
- 5.1.4. Lung Cancer
- 5.1.5. Pancreatic Cancer
- 5.1.6. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Solution Phase Synthesis (SPS)
- 5.2.2. Solid Phase Peptide Synthesis (SPPS)
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Prostate Cancer
- 6.1.2. Breast Cancer
- 6.1.3. Gastric Cancer
- 6.1.4. Lung Cancer
- 6.1.5. Pancreatic Cancer
- 6.1.6. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Solution Phase Synthesis (SPS)
- 6.2.2. Solid Phase Peptide Synthesis (SPPS)
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Prostate Cancer
- 7.1.2. Breast Cancer
- 7.1.3. Gastric Cancer
- 7.1.4. Lung Cancer
- 7.1.5. Pancreatic Cancer
- 7.1.6. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Solution Phase Synthesis (SPS)
- 7.2.2. Solid Phase Peptide Synthesis (SPPS)
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Prostate Cancer
- 8.1.2. Breast Cancer
- 8.1.3. Gastric Cancer
- 8.1.4. Lung Cancer
- 8.1.5. Pancreatic Cancer
- 8.1.6. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Solution Phase Synthesis (SPS)
- 8.2.2. Solid Phase Peptide Synthesis (SPPS)
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Prostate Cancer
- 9.1.2. Breast Cancer
- 9.1.3. Gastric Cancer
- 9.1.4. Lung Cancer
- 9.1.5. Pancreatic Cancer
- 9.1.6. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Solution Phase Synthesis (SPS)
- 9.2.2. Solid Phase Peptide Synthesis (SPPS)
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Synthetic Peptide Vaccine Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Prostate Cancer
- 10.1.2. Breast Cancer
- 10.1.3. Gastric Cancer
- 10.1.4. Lung Cancer
- 10.1.5. Pancreatic Cancer
- 10.1.6. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Solution Phase Synthesis (SPS)
- 10.2.2. Solid Phase Peptide Synthesis (SPPS)
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Anergis
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Antigen Express
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Apitope Technology
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Artificial Cell Technologies
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Benovus Bio
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Celgene Corporation
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Immatics biotechnologies
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Merck
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 ImmunoCellular Therapeutics
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 ISA Pharmaceuticals
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Lytix Biopharma
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Spotlight Innovation
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 OncoTherapy Science
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Peptech
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Pharmasyntez
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Sumitomo Dainippon Pharma
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 TapImmune
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Targovax
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.1 Anergis
List of Figures
- Figure 1: Global Synthetic Peptide Vaccine Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global Synthetic Peptide Vaccine Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Synthetic Peptide Vaccine Revenue (billion), by Application 2025 & 2033
- Figure 4: North America Synthetic Peptide Vaccine Volume (K), by Application 2025 & 2033
- Figure 5: North America Synthetic Peptide Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Synthetic Peptide Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Synthetic Peptide Vaccine Revenue (billion), by Types 2025 & 2033
- Figure 8: North America Synthetic Peptide Vaccine Volume (K), by Types 2025 & 2033
- Figure 9: North America Synthetic Peptide Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Synthetic Peptide Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Synthetic Peptide Vaccine Revenue (billion), by Country 2025 & 2033
- Figure 12: North America Synthetic Peptide Vaccine Volume (K), by Country 2025 & 2033
- Figure 13: North America Synthetic Peptide Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Synthetic Peptide Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Synthetic Peptide Vaccine Revenue (billion), by Application 2025 & 2033
- Figure 16: South America Synthetic Peptide Vaccine Volume (K), by Application 2025 & 2033
- Figure 17: South America Synthetic Peptide Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Synthetic Peptide Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Synthetic Peptide Vaccine Revenue (billion), by Types 2025 & 2033
- Figure 20: South America Synthetic Peptide Vaccine Volume (K), by Types 2025 & 2033
- Figure 21: South America Synthetic Peptide Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Synthetic Peptide Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Synthetic Peptide Vaccine Revenue (billion), by Country 2025 & 2033
- Figure 24: South America Synthetic Peptide Vaccine Volume (K), by Country 2025 & 2033
- Figure 25: South America Synthetic Peptide Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Synthetic Peptide Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Synthetic Peptide Vaccine Revenue (billion), by Application 2025 & 2033
- Figure 28: Europe Synthetic Peptide Vaccine Volume (K), by Application 2025 & 2033
- Figure 29: Europe Synthetic Peptide Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Synthetic Peptide Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Synthetic Peptide Vaccine Revenue (billion), by Types 2025 & 2033
- Figure 32: Europe Synthetic Peptide Vaccine Volume (K), by Types 2025 & 2033
- Figure 33: Europe Synthetic Peptide Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Synthetic Peptide Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Synthetic Peptide Vaccine Revenue (billion), by Country 2025 & 2033
- Figure 36: Europe Synthetic Peptide Vaccine Volume (K), by Country 2025 & 2033
- Figure 37: Europe Synthetic Peptide Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Synthetic Peptide Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Synthetic Peptide Vaccine Revenue (billion), by Application 2025 & 2033
- Figure 40: Middle East & Africa Synthetic Peptide Vaccine Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Synthetic Peptide Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Synthetic Peptide Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Synthetic Peptide Vaccine Revenue (billion), by Types 2025 & 2033
- Figure 44: Middle East & Africa Synthetic Peptide Vaccine Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Synthetic Peptide Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Synthetic Peptide Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Synthetic Peptide Vaccine Revenue (billion), by Country 2025 & 2033
- Figure 48: Middle East & Africa Synthetic Peptide Vaccine Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Synthetic Peptide Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Synthetic Peptide Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Synthetic Peptide Vaccine Revenue (billion), by Application 2025 & 2033
- Figure 52: Asia Pacific Synthetic Peptide Vaccine Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Synthetic Peptide Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Synthetic Peptide Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Synthetic Peptide Vaccine Revenue (billion), by Types 2025 & 2033
- Figure 56: Asia Pacific Synthetic Peptide Vaccine Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Synthetic Peptide Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Synthetic Peptide Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Synthetic Peptide Vaccine Revenue (billion), by Country 2025 & 2033
- Figure 60: Asia Pacific Synthetic Peptide Vaccine Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Synthetic Peptide Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Synthetic Peptide Vaccine Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 4: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global Synthetic Peptide Vaccine Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 8: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 10: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global Synthetic Peptide Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 20: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 22: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global Synthetic Peptide Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Brazil Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Argentina Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 32: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 34: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Country 2020 & 2033
- Table 36: Global Synthetic Peptide Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 40: Germany Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: France Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: Italy Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Spain Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: Russia Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Benelux Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: Nordics Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 56: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 58: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global Synthetic Peptide Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 62: Turkey Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 64: Israel Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 66: GCC Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 68: North Africa Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 70: South Africa Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Application 2020 & 2033
- Table 74: Global Synthetic Peptide Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Types 2020 & 2033
- Table 76: Global Synthetic Peptide Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Synthetic Peptide Vaccine Revenue billion Forecast, by Country 2020 & 2033
- Table 78: Global Synthetic Peptide Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 79: China Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 80: China Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 82: India Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 84: Japan Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 86: South Korea Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 90: Oceania Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Synthetic Peptide Vaccine Revenue (billion) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Synthetic Peptide Vaccine Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Synthetic Peptide Vaccine?
The projected CAGR is approximately 10.81%.
2. Which companies are prominent players in the Synthetic Peptide Vaccine?
Key companies in the market include Anergis, Antigen Express, Apitope Technology, Artificial Cell Technologies, Benovus Bio, Celgene Corporation, Immatics biotechnologies, Merck, ImmunoCellular Therapeutics, ISA Pharmaceuticals, Lytix Biopharma, Spotlight Innovation, OncoTherapy Science, Peptech, Pharmasyntez, Sumitomo Dainippon Pharma, TapImmune, Targovax.
3. What are the main segments of the Synthetic Peptide Vaccine?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 7.77 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Synthetic Peptide Vaccine," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Synthetic Peptide Vaccine report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Synthetic Peptide Vaccine?
To stay informed about further developments, trends, and reports in the Synthetic Peptide Vaccine, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
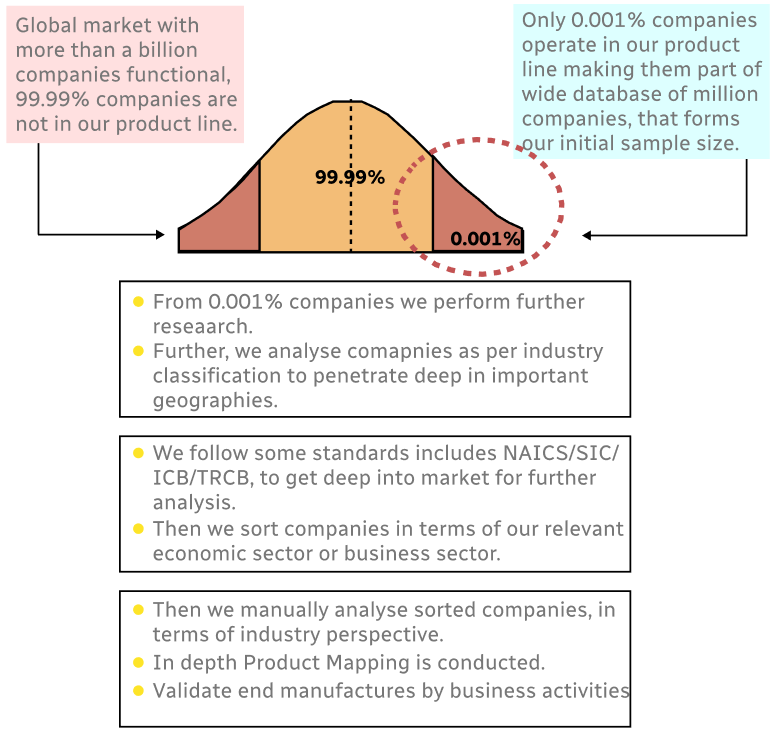
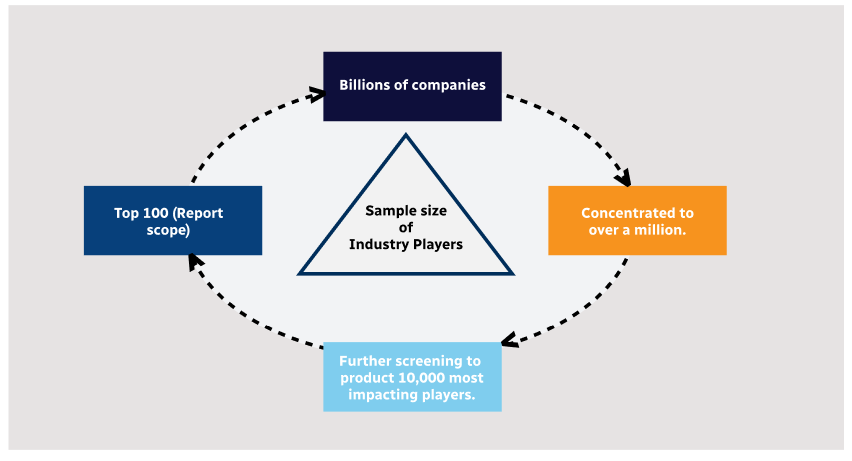
Step 1 - Identification of Relevant Samples Size from Population Database
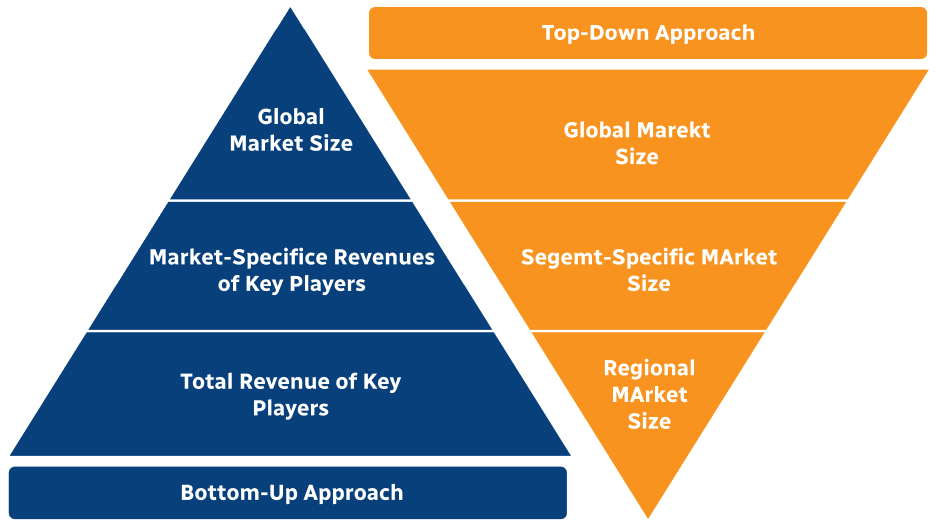
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
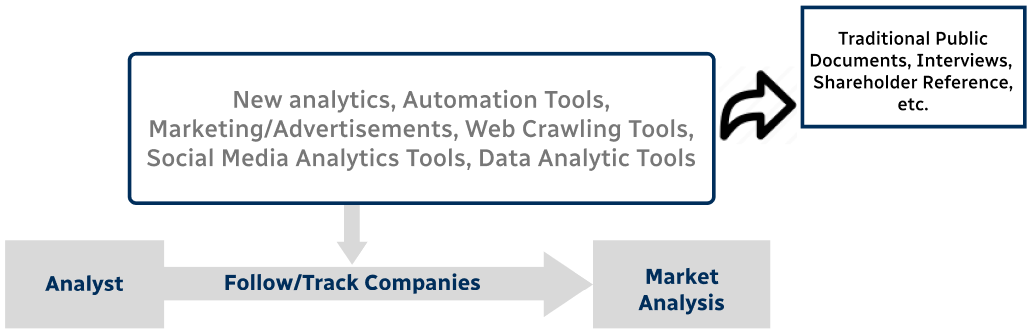
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


