Key Insights
The global Ethylene Oxide (EtO) Device Sterilization market is poised for robust growth, estimated to reach $3.5 billion in 2025. Driven by an increasing demand for sterile medical devices and stringent regulatory requirements for infection control, the market is projected to expand at a Compound Annual Growth Rate (CAGR) of 6% from 2025 to 2033. This growth is primarily fueled by the critical role of EtO sterilization in processing heat-sensitive and complex medical instruments that cannot withstand high temperatures or moisture. The rising prevalence of chronic diseases, coupled with the continuous innovation in medical device technology, further accentuates the need for effective and reliable sterilization methods, thereby propelling market expansion. Key applications within this market include hospitals and outpatient facilities, with a growing emphasis on contract sterilization services as healthcare providers increasingly outsource their sterilization needs to specialized providers.
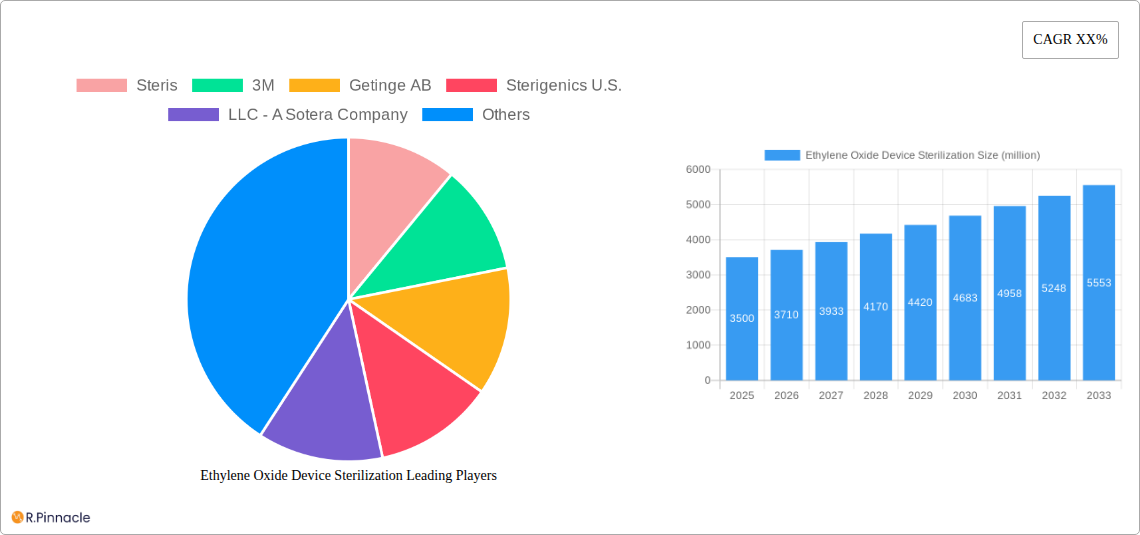
Ethylene Oxide Device Sterilization Market Size (In Billion)

The market's trajectory is shaped by several influencing factors. Advancements in EtO sterilization equipment, including enhanced safety features and improved efficiency, are contributing to market adoption. Furthermore, the growing healthcare infrastructure in emerging economies, particularly in the Asia Pacific region, presents significant growth opportunities. However, the market faces certain restraints, including increasing environmental regulations concerning EtO emissions and the development of alternative sterilization technologies. Despite these challenges, the inherent advantages of EtO sterilization, such as its broad material compatibility and deep penetration capabilities, ensure its continued relevance and demand. Leading companies such as Steris, 3M, and Getinge AB are actively investing in research and development, offering innovative solutions and expanding their global presence to cater to the evolving market landscape.
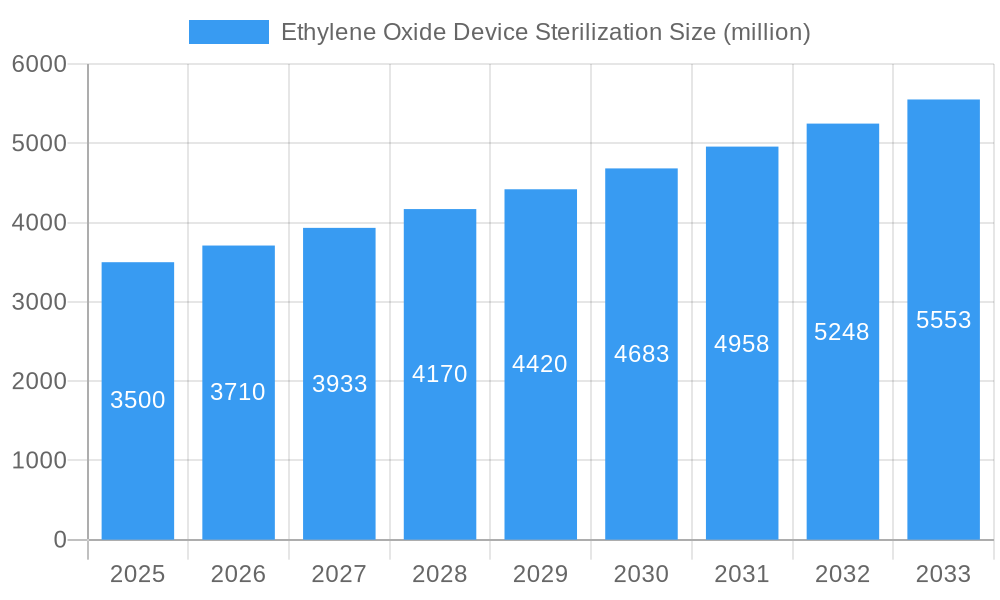
Ethylene Oxide Device Sterilization Company Market Share

Here is an SEO-optimized, reader-centric report description for Ethylene Oxide Device Sterilization:
This in-depth report provides a definitive analysis of the global Ethylene Oxide (EtO) Device Sterilization market, encompassing historical trends, current dynamics, and future projections. With a study period spanning from 2019 to 2033, this report offers invaluable insights for stakeholders seeking to understand market concentration, innovation drivers, regulatory landscapes, and competitive strategies. We delve into the intricate segmentation of the market by application and sterilization type, identifying key growth accelerators and emerging opportunities within this critical healthcare segment. Expected to reach a market value of $XX billion by 2033, with a Compound Annual Growth Rate (CAGR) of XX% during the forecast period (2025-2033), this research is essential for medical device manufacturers, contract sterilization providers, healthcare institutions, and investors.
Ethylene Oxide Device Sterilization Market Structure & Innovation Trends
The Ethylene Oxide Device Sterilization market exhibits a moderately concentrated structure, characterized by a blend of established global players and specialized regional providers. Innovation is primarily driven by the demand for enhanced sterilization efficacy, reduced cycle times, and the development of environmentally conscious EtO sterilization technologies. Regulatory frameworks, particularly concerning emissions and safety standards, play a pivotal role in shaping market dynamics and influencing R&D investments. The threat of product substitutes, such as steam sterilization or radiation, is moderate, with EtO retaining its dominance for heat-sensitive and complex medical devices. End-user demographics, predominantly hospitals and outpatient facilities, are increasingly prioritizing patient safety and regulatory compliance. Mergers and Acquisitions (M&A) activities have been observed, with deal values reaching $XX billion in the historical period (2019-2024), indicating strategic consolidation and market expansion. Key market share leaders are consistently investing in optimizing their sterilization processes and expanding their service offerings to meet growing global demand.
Ethylene Oxide Device Sterilization Market Dynamics & Trends
The Ethylene Oxide Device Sterilization market is experiencing robust growth, fueled by a confluence of critical factors. The escalating global prevalence of healthcare-associated infections (HAIs) and the increasing adoption of advanced medical devices that necessitate reliable sterilization methods are paramount growth drivers. Technological advancements are continuously refining EtO sterilization processes, leading to improved efficiency, reduced environmental impact, and enhanced compatibility with a wider range of medical materials. Consumer preferences, particularly from healthcare providers, lean towards validated, safe, and cost-effective sterilization solutions that guarantee aseptic conditions and patient well-being. The competitive landscape is dynamic, with key players focusing on innovation, capacity expansion, and strategic partnerships to secure market share. Market penetration is steadily increasing, driven by stringent regulatory requirements for medical device sterilization and a growing awareness of the importance of sterile instruments. The market is projected to experience sustained growth due to the indispensable role of EtO in sterilizing complex and heat-sensitive medical equipment, alongside the continuous innovation in sterilization equipment and processes that enhance safety and sustainability. Furthermore, the expanding global healthcare infrastructure and the increasing demand for minimally invasive surgical procedures, which often utilize sophisticated, sterilizable instruments, contribute significantly to market expansion. The commitment from leading companies to invest in research and development for more environmentally friendly EtO sterilization techniques is also a key trend, addressing regulatory pressures and corporate social responsibility initiatives. The continued reliance on EtO for a vast array of medical devices, from single-use instruments to complex implantable devices, underscores its enduring importance in the healthcare ecosystem.
Dominant Regions & Segments in Ethylene Oxide Device Sterilization
The North America region stands as a dominant force in the Ethylene Oxide Device Sterilization market. This dominance is propelled by several key drivers, including:
- Robust Healthcare Infrastructure: Extensive hospital networks, advanced research facilities, and a high patient volume necessitate consistent and reliable sterilization services.
- Strict Regulatory Oversight: The Food and Drug Administration (FDA) in the United States enforces rigorous standards for medical device sterilization, driving demand for compliant EtO services.
- Technological Adoption: Early and widespread adoption of advanced sterilization technologies and equipment ensures efficient and effective sterilization processes.
- Presence of Key Market Players: The region hosts major EtO sterilization service providers and equipment manufacturers, fostering innovation and competition.
- Economic Policies: Favorable healthcare spending and government initiatives supporting medical device manufacturing contribute to market growth.
Within this dominant region, the Hospitals application segment is a primary driver of EtO sterilization demand. Hospitals require a continuous supply of sterile instruments for a wide range of surgical procedures, diagnostic tests, and patient care. The increasing complexity of surgical tools and the need for sterilization of heat-sensitive materials further bolster the demand for EtO.
The Contract Sterilization Service type is also exceptionally prominent. Healthcare facilities often outsource sterilization to specialized contract providers to ensure compliance, manage costs, and maintain focus on patient care. These service providers possess the expertise, specialized equipment, and regulatory know-how to deliver reliable EtO sterilization solutions.
Ethylene Oxide Device Sterilization Product Innovations
Recent product innovations in Ethylene Oxide Device Sterilization focus on enhancing sterilization efficiency, reducing environmental impact, and improving material compatibility. Advancements include the development of low-temperature EtO sterilization systems, which are ideal for heat-sensitive medical devices. Automated control systems and real-time monitoring technologies are being integrated to ensure consistent and validated sterilization cycles. Furthermore, research into alternative sterilant formulations and containment technologies aims to minimize EtO emissions, addressing regulatory concerns and promoting sustainability. These innovations offer competitive advantages by providing safer, more efficient, and environmentally responsible sterilization solutions for a growing range of medical applications.
Report Scope & Segmentation Analysis
This report meticulously segments the Ethylene Oxide Device Sterilization market across key areas. The Application segment is divided into:
- Hospitals: This segment represents a significant portion of the market due to the high volume of sterilization needs for surgical instruments, implants, and other medical devices used in inpatient and outpatient settings. Growth is driven by increasing surgical procedures and a focus on infection control.
- Outpatient Facilities: This segment includes clinics and specialized centers that require sterilized medical devices for diagnostic and therapeutic purposes. Its growth is linked to the expansion of ambulatory care and the shift towards outpatient procedures.
- Others: This encompasses research laboratories, veterinary clinics, and other specialized healthcare settings that utilize EtO sterilized devices.
The Type segmentation includes:
- Contract Sterilization Service: This segment comprises third-party sterilization providers offering EtO sterilization as a service. It is expected to grow robustly due to its cost-effectiveness and expertise for manufacturers and healthcare providers.
- Sterilization Validation Service: This segment focuses on the critical process of validating EtO sterilization cycles to ensure efficacy and compliance. Its growth is directly tied to regulatory requirements and the need for robust quality assurance.
Key Drivers of Ethylene Oxide Device Sterilization Growth
The Ethylene Oxide Device Sterilization market is propelled by several key drivers. The persistent need for sterilizing heat-sensitive and complex medical devices, such as electronics, plastics, and intricate surgical tools, where alternatives like autoclaving are unsuitable, is a fundamental driver. Stringent regulatory mandates from bodies like the FDA and EMA, emphasizing the absolute requirement for sterile medical devices to prevent infections, further bolster demand. Technological advancements leading to more efficient, faster, and environmentally responsible EtO sterilization technologies are also crucial. The increasing global aging population and the rise in chronic diseases necessitate more medical procedures and, consequently, a greater demand for sterilized medical supplies. Finally, the expanding global healthcare infrastructure, particularly in emerging economies, contributes significantly to the growth of EtO sterilization services.
Challenges in the Ethylene Oxide Device Sterilization Sector
Despite its critical role, the Ethylene Oxide Device Sterilization sector faces significant challenges. Chief among these are stringent and evolving regulatory pressures concerning EtO emissions, leading to increased operational costs for compliance and potential facility closures or restrictions. Supply chain disruptions, particularly for EtO gas and related consumables, can impact service availability and pricing. Furthermore, public perception and environmental concerns surrounding EtO emissions necessitate continuous investment in emission control technologies and advocacy for its safe use. Competitive pressures from alternative sterilization methods, though limited for certain device types, remain a factor. Quantifiable impacts include increased capital expenditure for emission abatement systems and potential delays in sterilization services due to regulatory scrutiny.
Emerging Opportunities in Ethylene Oxide Device Sterilization
Emerging opportunities in the Ethylene Oxide Device Sterilization market are centered on innovation and sustainability. The development and adoption of advanced EtO sterilization technologies that significantly reduce emissions, such as enclosed loop systems and catalytic converters, present a major growth avenue. Expansion into emerging markets in Asia-Pacific and Latin America, where healthcare infrastructure is rapidly developing and demand for medical devices is rising, offers substantial untapped potential. The increasing complexity and sensitivity of new medical devices, particularly in fields like robotics and advanced prosthetics, will continue to rely on EtO's unique sterilization capabilities. Furthermore, offering comprehensive validation and consulting services alongside sterilization provides value-added opportunities for market players.
Leading Players in the Ethylene Oxide Device Sterilization Market
- Steris
- 3M
- Getinge AB
- Sterigenics U.S., LLC - A Sotera Company
- Andersen Sterilizers
- Ionisos
- Cosmed Group
- Sterility Equipment India Private Limited
Key Developments in Ethylene Oxide Device Sterilization Industry
- 2023/October: Launch of new low-emission EtO sterilization chamber technologies by leading equipment manufacturers, aimed at meeting stricter environmental regulations.
- 2023/July: Increased investment in R&D for alternative sterilant formulations to reduce reliance on traditional EtO gas.
- 2022/November: Several contract sterilization providers expanded their capacity to meet growing demand, particularly in North America and Europe.
- 2021/March: Regulatory bodies in key markets announced updated guidelines for EtO emissions, prompting industry-wide adjustments and technological upgrades.
- 2020/September: Strategic partnerships formed between medical device manufacturers and sterilization service providers to ensure supply chain resilience.
- 2019/December: Introduction of advanced monitoring and control systems for EtO sterilization processes to enhance validation and compliance.
Future Outlook for Ethylene Oxide Device Sterilization Market
The future outlook for the Ethylene Oxide Device Sterilization market remains exceptionally strong, driven by the indispensable nature of EtO for a vast array of critical medical devices. Anticipated growth accelerators include the continuous evolution of medical technology, leading to more complex and heat-sensitive instruments that demand EtO's unique sterilization capabilities. Strategic opportunities lie in the development and widespread adoption of next-generation EtO sterilization technologies that offer enhanced sustainability and reduced environmental impact. Expansion into rapidly developing healthcare markets globally presents significant untapped potential. The increasing global focus on patient safety and infection prevention will continue to underscore the vital role of reliable and validated EtO sterilization services, ensuring robust demand for the foreseeable future.
Ethylene Oxide Device Sterilization Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Outpatient Facilities
- 1.3. Others
-
2. Types
- 2.1. Contract Sterilization Service
- 2.2. Sterilization Validation Service
Ethylene Oxide Device Sterilization Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
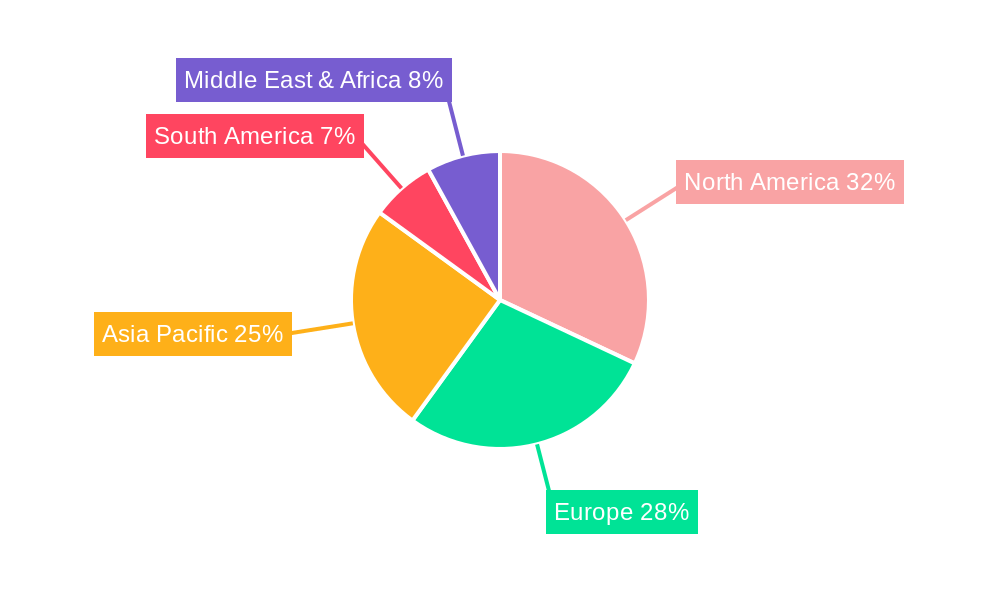
Ethylene Oxide Device Sterilization Regional Market Share

Geographic Coverage of Ethylene Oxide Device Sterilization
Ethylene Oxide Device Sterilization REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Outpatient Facilities
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Contract Sterilization Service
- 5.2.2. Sterilization Validation Service
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Outpatient Facilities
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Contract Sterilization Service
- 6.2.2. Sterilization Validation Service
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Outpatient Facilities
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Contract Sterilization Service
- 7.2.2. Sterilization Validation Service
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Outpatient Facilities
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Contract Sterilization Service
- 8.2.2. Sterilization Validation Service
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Outpatient Facilities
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Contract Sterilization Service
- 9.2.2. Sterilization Validation Service
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Outpatient Facilities
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Contract Sterilization Service
- 10.2.2. Sterilization Validation Service
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Ethylene Oxide Device Sterilization Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospitals
- 11.1.2. Outpatient Facilities
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Contract Sterilization Service
- 11.2.2. Sterilization Validation Service
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Steris
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 3M
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Getinge AB
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Sterigenics U.S.
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 LLC - A Sotera Company
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Andersen Sterilizers
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Ionisos
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Cosmed Group
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Sterility Equipment India Private Limited
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.1 Steris
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Ethylene Oxide Device Sterilization Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Ethylene Oxide Device Sterilization Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Ethylene Oxide Device Sterilization Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Ethylene Oxide Device Sterilization Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Ethylene Oxide Device Sterilization Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Ethylene Oxide Device Sterilization Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Ethylene Oxide Device Sterilization Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Ethylene Oxide Device Sterilization Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Ethylene Oxide Device Sterilization Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Ethylene Oxide Device Sterilization Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Ethylene Oxide Device Sterilization Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Ethylene Oxide Device Sterilization Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Ethylene Oxide Device Sterilization Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Ethylene Oxide Device Sterilization Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Ethylene Oxide Device Sterilization Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Ethylene Oxide Device Sterilization Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Ethylene Oxide Device Sterilization Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Ethylene Oxide Device Sterilization Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Ethylene Oxide Device Sterilization Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Ethylene Oxide Device Sterilization Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Ethylene Oxide Device Sterilization Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Ethylene Oxide Device Sterilization Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Ethylene Oxide Device Sterilization Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Ethylene Oxide Device Sterilization Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Ethylene Oxide Device Sterilization Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Ethylene Oxide Device Sterilization Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Ethylene Oxide Device Sterilization Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Ethylene Oxide Device Sterilization Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Ethylene Oxide Device Sterilization Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Ethylene Oxide Device Sterilization Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Ethylene Oxide Device Sterilization Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Ethylene Oxide Device Sterilization Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Ethylene Oxide Device Sterilization Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Ethylene Oxide Device Sterilization?
The projected CAGR is approximately 13.4%.
2. Which companies are prominent players in the Ethylene Oxide Device Sterilization?
Key companies in the market include Steris, 3M, Getinge AB, Sterigenics U.S., LLC - A Sotera Company, Andersen Sterilizers, Ionisos, Cosmed Group, Sterility Equipment India Private Limited.
3. What are the main segments of the Ethylene Oxide Device Sterilization?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Ethylene Oxide Device Sterilization," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Ethylene Oxide Device Sterilization report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Ethylene Oxide Device Sterilization?
To stay informed about further developments, trends, and reports in the Ethylene Oxide Device Sterilization, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
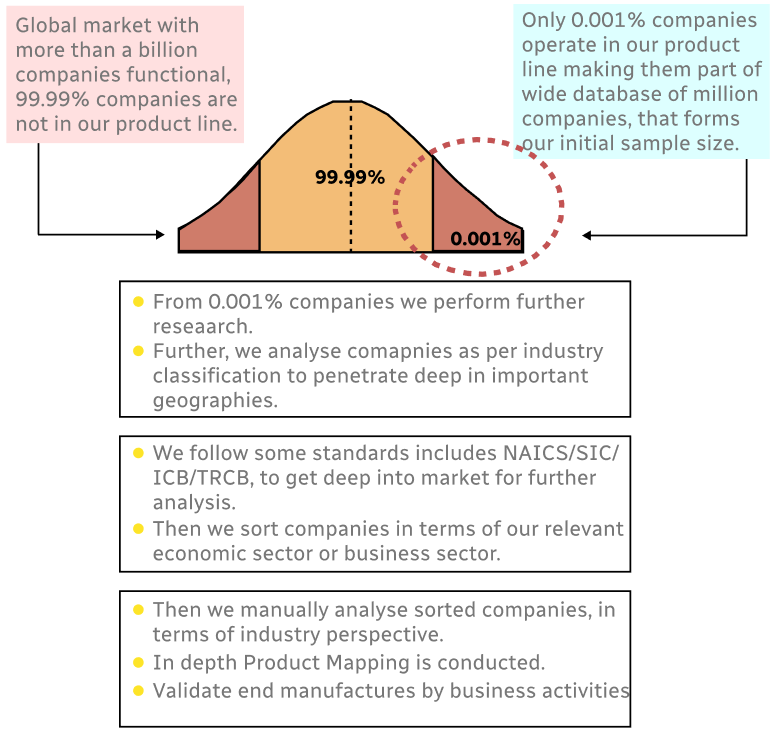
Step 1 - Identification of Relevant Samples Size from Population Database
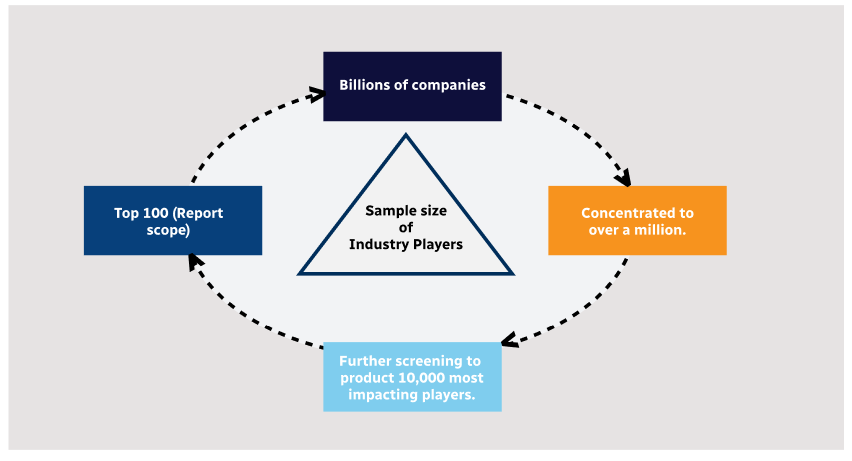
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
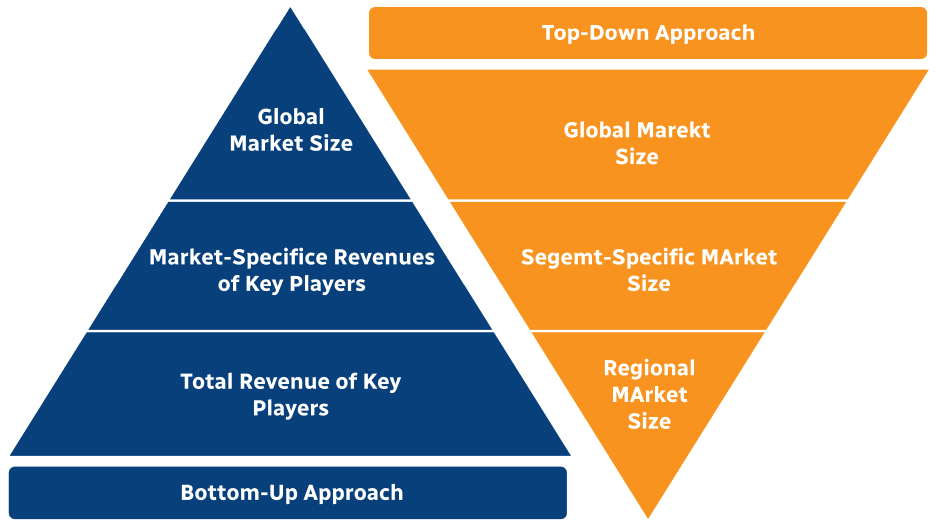
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
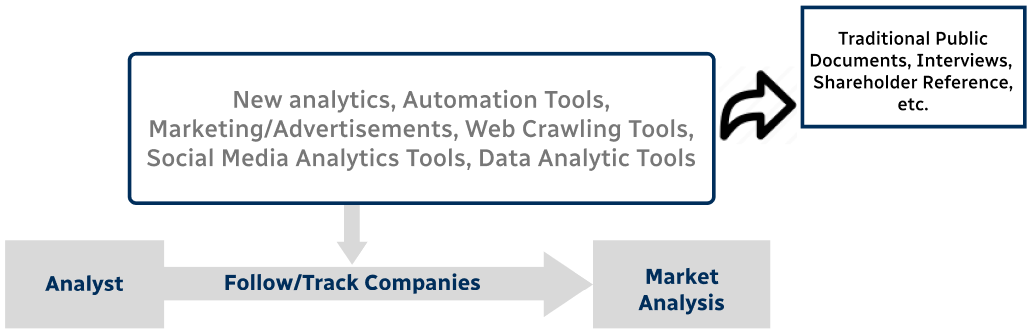
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


