Key Insights
The Mexico In-Vitro Diagnostics (IVD) Industry is poised for significant expansion, projected to reach a market size of $2.07 Billion by 2025, demonstrating robust growth at a Compound Annual Growth Rate (CAGR) of 7.18% throughout the forecast period. This upward trajectory is fueled by several key drivers, including an increasing prevalence of infectious diseases and chronic conditions like cancer, diabetes, and autoimmune disorders, necessitating more frequent and accurate diagnostic testing. Advancements in diagnostic technologies, particularly in molecular diagnostics and immunoassays, are enhancing sensitivity and specificity, thereby driving adoption. The growing demand for rapid and point-of-care testing solutions, alongside an expanding healthcare infrastructure and increased government initiatives focused on public health and disease surveillance, further bolster market growth. The rising disposable incomes and growing health consciousness among the Mexican population also contribute to a greater willingness to invest in advanced diagnostic services.
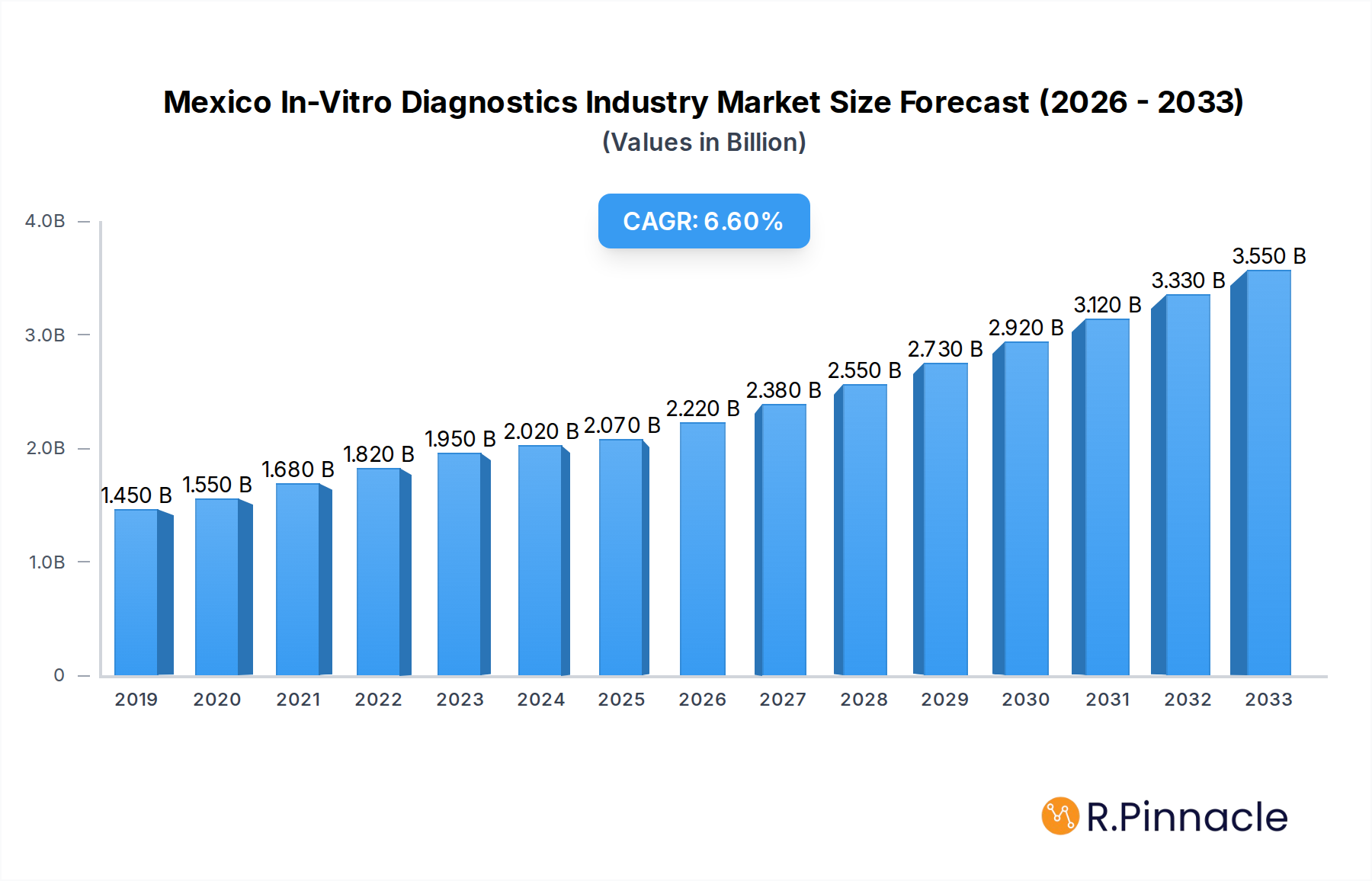
Mexico In-Vitro Diagnostics Industry Market Size (In Billion)

The IVD market in Mexico is segmented across various techniques, products, usability, applications, and end-users, reflecting the diverse needs of the healthcare ecosystem. Clinical chemistry and immunoassays represent dominant techniques, while instruments and reagents form the core product segments. Disposable IVD devices are gaining traction due to convenience and reduced risk of contamination, particularly in the context of infectious disease outbreaks. Key applications driving growth include infectious diseases, cancer, cardiology, and diabetes, reflecting the country's major health challenges. Diagnostic laboratories and hospitals and clinics are the primary end-users, leveraging these advanced diagnostics to improve patient outcomes. Despite the promising outlook, the market faces restraints such as the high cost of advanced IVD technologies and reagents, a shortage of skilled healthcare professionals trained in operating sophisticated diagnostic equipment, and complex regulatory pathways for new product approvals. However, the continuous innovation by leading companies and the strategic focus on emerging markets like Mexico are expected to mitigate these challenges and unlock substantial opportunities.
Mexico In-Vitro Diagnostics Industry Company Market Share

Mexico In-Vitro Diagnostics Industry: Market Analysis and Forecast 2019-2033
This comprehensive report delves into the dynamic Mexico In-Vitro Diagnostics (IVD) industry, offering an in-depth analysis of market structure, key trends, and future outlook. Spanning a study period from 2019 to 2033, with a base and estimated year of 2025 and a forecast period from 2025 to 2033, this report provides actionable insights for industry stakeholders. We meticulously examine market segmentation, growth drivers, challenges, and emerging opportunities, all while leveraging high-ranking keywords to ensure maximum search visibility for industry professionals seeking in-vitro diagnostics market size, Mexico healthcare, molecular diagnostics Mexico, and diagnostic laboratory trends.
Mexico In-Vitro Diagnostics Industry Market Structure & Innovation Trends
The Mexico In-Vitro Diagnostics industry exhibits a moderately consolidated market structure, with leading players like Abbott Laboratories, Siemens Healthineers AG, and F Hoffmann-La Roche AG holding significant market shares. Innovation is a key driver, fueled by advancements in molecular diagnostics and an increasing demand for point-of-care testing solutions. Regulatory frameworks, primarily overseen by the Mexican regulatory agencies, are evolving to accommodate novel technologies and ensure product safety and efficacy. Substitutes for traditional IVD methods are emerging, particularly in the realm of advanced imaging and genetic sequencing, posing a gradual competitive threat. End-user demographics are shifting towards an aging population with a higher prevalence of chronic diseases, influencing product demand. Mergers and acquisitions (M&A) activity remains a strategic tool for market expansion and portfolio diversification, with deal values potentially reaching into the hundreds of millions for significant acquisitions.
Mexico In-Vitro Diagnostics Industry Market Dynamics & Trends
The Mexico In-Vitro Diagnostics industry is poised for robust growth, projected to experience a Compound Annual Growth Rate (CAGR) of approximately 7.5% during the forecast period. This expansion is primarily driven by several converging factors. Firstly, an increasing prevalence of infectious diseases, coupled with a growing awareness and focus on early detection of chronic conditions such as cancer, diabetes, and cardiovascular diseases, is creating sustained demand for diagnostic testing. The Mexican government's ongoing investments in healthcare infrastructure and public health initiatives further bolster market penetration. Technological disruptions are at the forefront of this growth, with significant advancements in molecular diagnostics, including PCR and next-generation sequencing, enabling more accurate and faster disease identification. The rise of liquid biopsy techniques for cancer diagnosis represents a major innovation. Consumer preferences are increasingly leaning towards less invasive, faster, and more convenient diagnostic solutions, including at-home testing kits and point-of-care devices. This is pushing manufacturers to develop user-friendly and accessible products. Competitive dynamics are characterized by intense rivalry among global giants and emerging local players, with a focus on product differentiation, strategic partnerships, and market expansion. The growing demand for personalized medicine is also shaping market trends, necessitating the development of highly specific and sensitive diagnostic tools. Economic stability and increasing disposable incomes in certain segments of the Mexican population contribute to a greater willingness to invest in advanced healthcare diagnostics. The digital transformation of healthcare, including the adoption of laboratory information systems (LIS) and electronic health records (EHRs), is also streamlining diagnostic workflows and enhancing data management, further propelling market growth.
Dominant Regions & Segments in Mexico In-Vitro Diagnostics Industry
The Central Region of Mexico, encompassing major metropolitan areas like Mexico City and Guadalajara, stands out as the dominant region in the in-vitro diagnostics industry. This dominance is attributed to a higher concentration of advanced healthcare facilities, a larger patient population, and a robust network of diagnostic laboratories.
Key Drivers for Regional Dominance:
- Economic Hub: Central Mexico represents the economic powerhouse of the country, attracting significant investment in healthcare infrastructure.
- Population Density: The region hosts a substantial portion of Mexico's population, leading to a higher demand for diagnostic services.
- Healthcare Infrastructure: A greater number of well-equipped hospitals, specialized clinics, and independent diagnostic laboratories are concentrated here.
- Skilled Workforce: Access to a skilled pool of healthcare professionals, including pathologists, laboratory technicians, and researchers, supports the advanced use of IVD technologies.
Analyzing the Technique segmentation, Molecular Diagnostics is emerging as a leading segment, driven by its precision and versatility in identifying infectious diseases, genetic disorders, and various cancers. The COVID-19 pandemic significantly accelerated the adoption and demand for molecular diagnostic tests, a trend that is expected to continue.
- Molecular Diagnostics Dominance: Its ability to detect pathogens at very low concentrations, coupled with the growing understanding of genetic predispositions to diseases, makes it indispensable. The development of rapid diagnostic tests for infectious agents and advancements in genetic testing for oncology and rare diseases are key contributors.
In terms of Product segmentation, Reagents consistently represent the largest market share due to their consumable nature and the need for continuous replenishment for a wide range of diagnostic assays.
- Reagent Market Strength: The broad application of reagents across various diagnostic techniques, from routine clinical chemistry to highly specialized immunoassays, ensures their sustained demand.
For Usability, Disposable IVD Devices are gaining prominence, driven by their convenience, sterility assurance, and the increasing preference for single-use solutions in infection control and personalized medicine.
- Disposable Device Growth: The emphasis on preventing cross-contamination and the development of user-friendly point-of-care devices contribute to the growing adoption of disposables.
Regarding Application, Infectious Disease testing remains a critical and dominant application, amplified by ongoing concerns regarding public health and the potential for outbreaks.
- Infectious Disease Focus: The continuous need for rapid and accurate diagnosis of viral, bacterial, and fungal infections drives significant market activity.
Among End Users, Diagnostic Laboratories represent the largest segment, as they are the primary providers of IVD testing services across the country.
- Laboratory Dominance: The proliferation of both large-scale reference laboratories and smaller, specialized diagnostic centers underscores their pivotal role in the IVD ecosystem.
Mexico In-Vitro Diagnostics Industry Product Innovations
Product innovations in the Mexico IVD industry are rapidly advancing, focusing on enhanced sensitivity, speed, and multiplexing capabilities. Molecular diagnostics are at the forefront, with the development of highly accurate PCR-based tests for infectious diseases and oncology markers. Immunoassays are seeing improvements in automation and detection technologies, offering faster turnaround times. The increasing demand for point-of-care testing (POCT) is driving the innovation of portable and user-friendly instruments and assay kits for decentralized testing environments. These innovations aim to improve patient outcomes by enabling earlier and more precise diagnoses, thereby offering a significant competitive advantage.
Report Scope & Segmentation Analysis
This report meticulously segments the Mexico In-Vitro Diagnostics industry across multiple dimensions to provide a granular market view. The Technique segmentation includes Clinical Chemistry, Molecular Diagnostics, Immuno Diagnostics, Hematology, and Other Techniques, each contributing distinct market values and growth projections based on their application and adoption rates. The Product segmentation covers Instruments, Reagents, and Other Products, with reagents projected to hold a significant market share due to their consumable nature. In terms of Usability, Disposable IVD Devices are expected to witness robust growth, driven by convenience and infection control. The Application segmentation analyzes demand across Infectious Disease, Cancer/Oncology, Cardiology, Autoimmune Disease, Diabetes, Nephrology, and Other Applications, with Infectious Disease and Oncology anticipated to be major growth areas. Finally, the End Users are categorized into Diagnostic Laboratories, Hospitals and Clinics, and Other End Users, with diagnostic laboratories forming the largest market segment.
Key Drivers of Mexico In-Vitro Diagnostics Industry Growth
Several factors are propelling the growth of the Mexico In-Vitro Diagnostics industry. A rising incidence of chronic diseases like diabetes and cancer, coupled with an aging population, significantly increases the demand for diagnostic testing. Government initiatives focused on improving healthcare access and infrastructure, including investments in public health programs and the expansion of laboratory services, are critical accelerators. Technological advancements, particularly in molecular diagnostics and automation, are enhancing the accuracy, speed, and accessibility of IVD tests. Furthermore, increased health awareness among the population and a growing demand for personalized medicine are contributing to market expansion. The competitive landscape, with global players investing in the region, also drives innovation and market development.
Challenges in the Mexico In-Vitro Diagnostics Industry Sector
Despite its growth potential, the Mexico IVD industry faces several challenges. Regulatory hurdles and the time-consuming approval processes for new diagnostic technologies can slow down market entry. Inadequate healthcare infrastructure in remote or less developed regions can limit access to advanced diagnostic services. The high cost of certain advanced IVD instruments and reagents can be a barrier for some healthcare providers and patients, especially in public healthcare settings. Furthermore, supply chain disruptions, particularly for imported raw materials and finished products, can impact availability and pricing. Intense competition from established global players and the need for continuous investment in research and development to keep pace with technological advancements also present significant challenges.
Emerging Opportunities in Mexico In-Vitro Diagnostics Industry
The Mexico IVD industry is ripe with emerging opportunities. The growing demand for point-of-care testing (POCT) presents a significant avenue for growth, enabling faster diagnoses in primary care settings and remote areas. The increasing focus on personalized medicine and companion diagnostics offers substantial potential for niche market development, particularly in oncology. The expansion of telehealth services creates opportunities for remote diagnostic monitoring and the integration of IVD data into digital health platforms. Furthermore, collaborations between IVD manufacturers and academic institutions for research and development can foster innovation. There is also a growing opportunity in developing cost-effective and accessible diagnostic solutions tailored to the specific needs of the Mexican population.
Leading Players in the Mexico In-Vitro Diagnostics Industry Market
- Abbott Laboratories
- Siemens Healthineers AG
- F Hoffmann-La Roche AG
- Thermo Fischer Scientific Inc
- Becton Dickinson and Company
- Bio-Rad Laboratories Inc
- BioMerieux
- Ortho Clinical Diagnostics
- Sysmex Corporation
- Danaher Corporation
- Seegene Inc
- Co-Diagnostics Inc
Key Developments in Mexico In-Vitro Diagnostics Industry Industry
- October 2020: BioMarin and Perkin Elmer partnered to support BioMarin's "Support Testing Program" for Mexico patients, providing biochemical and molecular genetic testing for suspected Batten disease or Mucopolysaccharidosis (MPS) conditions.
- May 2020: Co-Diagnostics Inc. received approval for sale from the Mexican Department of Epidemiology for its product "Logix Smart Coronavirus COVID-19 Test" in Mexico.
Future Outlook for Mexico In-Vitro Diagnostics Industry Market
The future outlook for the Mexico In-Vitro Diagnostics industry is exceptionally positive, driven by sustained investments in healthcare modernization and a proactive approach to public health. The increasing adoption of advanced technologies like AI-powered diagnostics, liquid biopsies, and expanded molecular testing will revolutionize disease detection and management. Growth accelerators include the continued expansion of molecular diagnostics for infectious diseases and oncology, alongside a surge in demand for point-of-care solutions and home-use diagnostic devices. The strategic focus on precision medicine and the integration of IVD data into comprehensive digital health ecosystems will further enhance market potential. Industry players can anticipate significant opportunities through strategic partnerships, product innovation, and market penetration into underserved regions, solidifying Mexico's position as a key market in the Latin American IVD landscape.
Mexico In-Vitro Diagnostics Industry Segmentation
-
1. Technique
- 1.1. Clinical Chemistry
- 1.2. Molecular Diagnostics
- 1.3. Immuno Diagnostics
- 1.4. Hematology
- 1.5. Other Techniques
-
2. Product
- 2.1. Instrument
- 2.2. Reagent
- 2.3. Other Products
-
3. Usability
- 3.1. Disposable IVD Devices
- 3.2. Reusable IVD Devices
-
4. Application
- 4.1. Infectious Disease
- 4.2. Cancer/Oncology
- 4.3. Cardiology
- 4.4. Autoimmune Disease
- 4.5. Diabetes
- 4.6. Nephrology
- 4.7. Other Applications
-
5. End Users
- 5.1. Diagnostic Laboratories
- 5.2. Hospitals and Clinics
- 5.3. Other End Users
Mexico In-Vitro Diagnostics Industry Segmentation By Geography
- 1. Mexico
Mexico In-Vitro Diagnostics Industry Regional Market Share

Geographic Coverage of Mexico In-Vitro Diagnostics Industry
Mexico In-Vitro Diagnostics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.18% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Technique
- 5.1.1. Clinical Chemistry
- 5.1.2. Molecular Diagnostics
- 5.1.3. Immuno Diagnostics
- 5.1.4. Hematology
- 5.1.5. Other Techniques
- 5.2. Market Analysis, Insights and Forecast - by Product
- 5.2.1. Instrument
- 5.2.2. Reagent
- 5.2.3. Other Products
- 5.3. Market Analysis, Insights and Forecast - by Usability
- 5.3.1. Disposable IVD Devices
- 5.3.2. Reusable IVD Devices
- 5.4. Market Analysis, Insights and Forecast - by Application
- 5.4.1. Infectious Disease
- 5.4.2. Cancer/Oncology
- 5.4.3. Cardiology
- 5.4.4. Autoimmune Disease
- 5.4.5. Diabetes
- 5.4.6. Nephrology
- 5.4.7. Other Applications
- 5.5. Market Analysis, Insights and Forecast - by End Users
- 5.5.1. Diagnostic Laboratories
- 5.5.2. Hospitals and Clinics
- 5.5.3. Other End Users
- 5.6. Market Analysis, Insights and Forecast - by Region
- 5.6.1. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Technique
- 6. Mexico In-Vitro Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Technique
- 6.1.1. Clinical Chemistry
- 6.1.2. Molecular Diagnostics
- 6.1.3. Immuno Diagnostics
- 6.1.4. Hematology
- 6.1.5. Other Techniques
- 6.2. Market Analysis, Insights and Forecast - by Product
- 6.2.1. Instrument
- 6.2.2. Reagent
- 6.2.3. Other Products
- 6.3. Market Analysis, Insights and Forecast - by Usability
- 6.3.1. Disposable IVD Devices
- 6.3.2. Reusable IVD Devices
- 6.4. Market Analysis, Insights and Forecast - by Application
- 6.4.1. Infectious Disease
- 6.4.2. Cancer/Oncology
- 6.4.3. Cardiology
- 6.4.4. Autoimmune Disease
- 6.4.5. Diabetes
- 6.4.6. Nephrology
- 6.4.7. Other Applications
- 6.5. Market Analysis, Insights and Forecast - by End Users
- 6.5.1. Diagnostic Laboratories
- 6.5.2. Hospitals and Clinics
- 6.5.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Technique
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Becton Dickinson and Company
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Bio-Rad Laboratories Inc
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Co-Diagnostics Inc
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 BioMerieux
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Seegene Inc
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Abbott Laboratories
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Ortho Clinical Diagnostics
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Siemens Healthineers AG
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Danaher Corporation
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 F Hoffmann-La Roche AG
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.11 Thermo Fischer Scientific Inc
- 7.1.11.1. Company Overview
- 7.1.11.2. Products
- 7.1.11.3. Company Financials
- 7.1.11.4. SWOT Analysis
- 7.1.12 Sysmex Corporation
- 7.1.12.1. Company Overview
- 7.1.12.2. Products
- 7.1.12.3. Company Financials
- 7.1.12.4. SWOT Analysis
- 7.1.1 Becton Dickinson and Company
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Mexico In-Vitro Diagnostics Industry Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: Mexico In-Vitro Diagnostics Industry Share (%) by Company 2025
List of Tables
- Table 1: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Technique 2020 & 2033
- Table 2: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Technique 2020 & 2033
- Table 3: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Product 2020 & 2033
- Table 4: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 5: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Usability 2020 & 2033
- Table 6: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Usability 2020 & 2033
- Table 7: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 8: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 9: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by End Users 2020 & 2033
- Table 10: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by End Users 2020 & 2033
- Table 11: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 12: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 13: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Technique 2020 & 2033
- Table 14: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Technique 2020 & 2033
- Table 15: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Product 2020 & 2033
- Table 16: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 17: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Usability 2020 & 2033
- Table 18: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Usability 2020 & 2033
- Table 19: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 20: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 21: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by End Users 2020 & 2033
- Table 22: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by End Users 2020 & 2033
- Table 23: Mexico In-Vitro Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 24: Mexico In-Vitro Diagnostics Industry Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Mexico In-Vitro Diagnostics Industry?
The projected CAGR is approximately 7.18%.
2. Which companies are prominent players in the Mexico In-Vitro Diagnostics Industry?
Key companies in the market include Becton Dickinson and Company, Bio-Rad Laboratories Inc, Co-Diagnostics Inc , BioMerieux, Seegene Inc, Abbott Laboratories, Ortho Clinical Diagnostics, Siemens Healthineers AG, Danaher Corporation, F Hoffmann-La Roche AG, Thermo Fischer Scientific Inc, Sysmex Corporation.
3. What are the main segments of the Mexico In-Vitro Diagnostics Industry?
The market segments include Technique, Product, Usability, Application, End Users.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.07 Million as of 2022.
5. What are some drivers contributing to market growth?
High Prevalence of Chronic Diseases; Growing Usage of Point-of-Care Diagnostics.
6. What are the notable trends driving market growth?
The Disposable IVD Devices Segment Dominates the Mexican In Vitro Diagnostics Market.
7. Are there any restraints impacting market growth?
Lack of Proper Reimbursement; Stringent Regulatory Framework.
8. Can you provide examples of recent developments in the market?
In October 2020, BioMarin and Perkin Elmer partnered to support BioMarin's "Support Testing Program" for Mexico patients. The company will be providing biochemical and molecular genetic testing for patients with suspicion of Batten disease or a Mucopolysaccharidosis (MPS) condition.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Mexico In-Vitro Diagnostics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Mexico In-Vitro Diagnostics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Mexico In-Vitro Diagnostics Industry?
To stay informed about further developments, trends, and reports in the Mexico In-Vitro Diagnostics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


