Key Insights
The European ovarian cancer diagnostics and therapeutics market is poised for significant expansion, driven by increasing disease prevalence, technological advancements in diagnostics, and the advent of targeted therapies. The market is projected to reach $7.62 billion by 2025, with a robust compound annual growth rate (CAGR) of 14.99% from 2025 to 2033. Key growth drivers include heightened awareness of ovarian cancer screening, enabling earlier detection and intervention, and continuous research and development yielding novel immunotherapies and personalized medicine approaches. Market segmentation encompasses epithelial ovarian tumors, germ cell tumors, and other subtypes, each with distinct diagnostic and therapeutic needs. Diagnostic modalities, particularly imaging, play a crucial role in improving patient outcomes. Leading markets include Germany, France, the United Kingdom, and Italy, owing to their large populations and sophisticated healthcare systems.
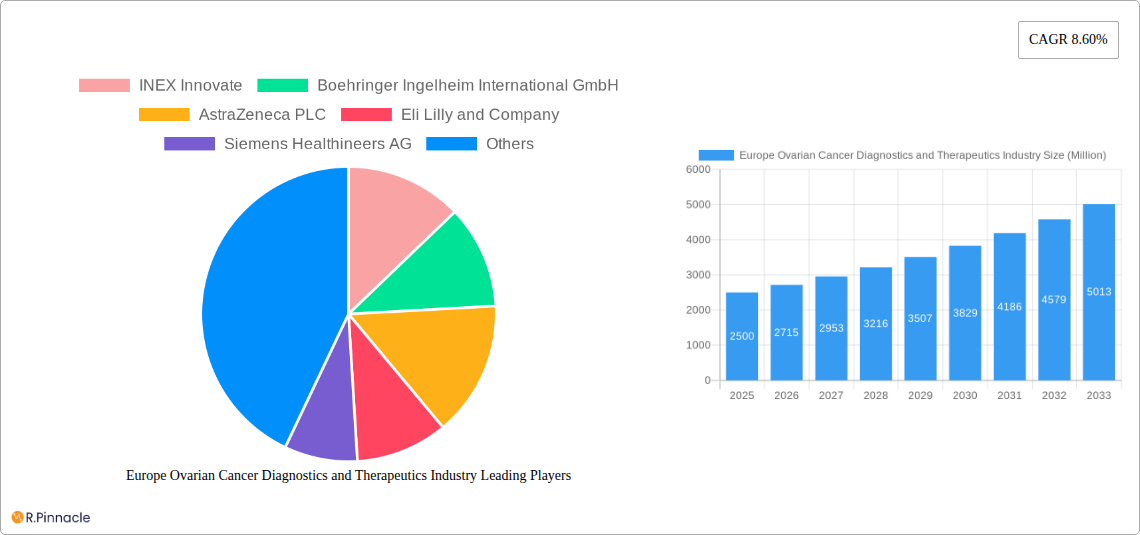
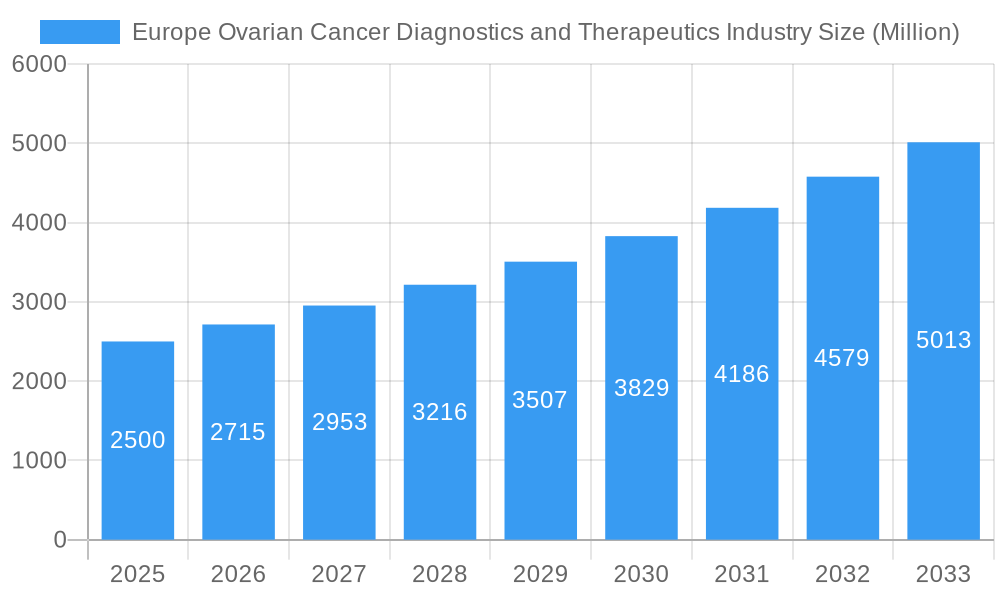
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Market Size (In Billion)

Despite market growth, challenges such as high treatment costs for advanced therapies and the common late-stage diagnosis of ovarian cancer, which impacts prognosis, persist. However, the development of enhanced diagnostic tools, targeted therapies, and public awareness campaigns are expected to overcome these obstacles and foster continued market growth. The competitive landscape features key players like Boehringer Ingelheim, AstraZeneca, Eli Lilly, and Siemens Healthineers, actively engaged in R&D and market penetration. The European market's strategic importance is underscored by its advanced healthcare infrastructure and the region's notable ovarian cancer prevalence.
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Company Market Share

This report offers a comprehensive analysis of the European ovarian cancer diagnostics and therapeutics industry, providing critical insights for stakeholders. Covering the forecast period from 2019 to 2033, with a specific focus on the 2025 base year, this study meticulously details market dynamics, competitive intelligence, and future growth trajectories.
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Market Structure & Innovation Trends
This section analyzes the market concentration, innovation drivers, regulatory frameworks, and competitive dynamics within the European ovarian cancer diagnostics and therapeutics market. We delve into market share distribution among key players, M&A activities, and the influence of regulatory changes on market growth. The report includes a detailed examination of the factors driving innovation, including technological advancements and the increasing demand for effective treatments.
- Market Concentration: The market exhibits a moderately concentrated structure, with a few major players holding significant market share (xx%). Smaller companies and start-ups play a vital role in innovation.
- Innovation Drivers: Technological advancements in diagnostics (e.g., liquid biopsies, advanced imaging) and targeted therapies are key innovation drivers. Increased research funding and collaborations further stimulate innovation.
- Regulatory Framework: Stringent regulatory approvals impact market entry and product launches, while also ensuring patient safety and efficacy. The report analyzes the influence of EMA (European Medicines Agency) guidelines.
- Product Substitutes: The availability of alternative treatment options influences market competition and pricing strategies.
- End-User Demographics: Analysis of patient demographics and treatment patterns provides insights into market segmentation and unmet needs.
- M&A Activities: The report examines completed and projected M&A deals, including deal values (xx Million) and their impact on market consolidation and innovation. Examples include potential acquisitions of smaller biotech companies by larger pharmaceutical firms.
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Market Dynamics & Trends
This section explores the key factors influencing the market's growth trajectory, including market size, CAGR, and market penetration rates. We analyze the impact of technological disruptions, evolving consumer preferences, and intense competition on market dynamics.
The European ovarian cancer diagnostics and therapeutics market is experiencing robust growth, driven by factors such as increasing prevalence of ovarian cancer, advancements in treatment modalities, and rising healthcare expenditure. The CAGR for the forecast period (2025-2033) is estimated at xx%. Market penetration of targeted therapies is steadily increasing, with expected penetration of xx% by 2033. Technological disruptions, such as the development of personalized medicine approaches, are transforming treatment strategies and creating new market opportunities. The competitive landscape is characterized by intense rivalry among established pharmaceutical companies and emerging biotech firms, leading to continuous innovation and price competition.
Dominant Regions & Segments in Europe Ovarian Cancer Diagnostics and Therapeutics Industry
This section identifies the leading regions and segments within the European market, analyzing their growth drivers and market dominance. We examine the performance of different cancer types (Epithelial Ovarian Tumors, Ovarian Germ Cell Tumors, Other Cancer Types) and modalities (Diagnosis, Therapeutics).
Leading Regions: [Germany, France, UK] are expected to dominate the market due to factors like higher healthcare spending, robust healthcare infrastructure, and well-established research ecosystems.
Leading Segments:
- Cancer Type: Epithelial Ovarian Tumors segment holds the largest market share, driven by its higher prevalence.
- Modality: The therapeutics segment dominates, reflecting the significant investment in drug development and the ongoing search for more effective treatments.
Key Drivers:
- Strong Healthcare Infrastructure: Developed healthcare systems in leading countries support early diagnosis and effective treatment.
- Government Initiatives & Funding: Government initiatives for cancer research and improved healthcare access further stimulate market growth.
- High Healthcare Expenditure: High per capita healthcare spending allows for greater access to advanced diagnostics and therapies.
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Product Innovations
The European ovarian cancer diagnostics and therapeutics market witnesses continuous product innovation. Recent advancements in targeted therapies, including PARP inhibitors and immunotherapy, have significantly improved treatment outcomes. Diagnostic tools, such as advanced imaging techniques and liquid biopsies, enable earlier and more precise diagnosis. This translates to better patient management and improved survival rates. The market is also witnessing the emergence of novel therapeutic approaches such as CAR T-cell therapy and oncolytic viruses, though these are still in earlier stages of development and market penetration.
Report Scope & Segmentation Analysis
This report segments the European ovarian cancer diagnostics and therapeutics market by cancer type (Epithelial Ovarian Tumors, Ovarian Germ Cell Tumors, Other Cancer Types) and modality (Diagnosis, Therapeutics). Each segment is analyzed in terms of market size (xx Million in 2025), growth projections (xx% CAGR), and competitive dynamics. The Epithelial Ovarian Tumors segment is projected to witness the highest growth due to its prevalence. The therapeutics segment is expected to account for a larger market share than the diagnostics segment, given the high demand for effective treatments.
Key Drivers of Europe Ovarian Cancer Diagnostics and Therapeutics Industry Growth
Several factors drive the growth of the European ovarian cancer diagnostics and therapeutics market. These include:
- Rising Prevalence of Ovarian Cancer: The increasing incidence of ovarian cancer fuels demand for better diagnostics and treatments.
- Technological Advancements: Innovations in targeted therapies, immunotherapies, and diagnostic tools are revolutionizing treatment approaches.
- Increased Healthcare Spending: Rising healthcare expenditure enables greater access to advanced treatments and diagnostic technologies.
- Favorable Regulatory Environment: Supportive regulatory frameworks facilitate the development and launch of new therapies.
Challenges in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry Sector
Despite significant growth potential, the European ovarian cancer diagnostics and therapeutics market faces challenges:
- High Drug Prices: The cost of advanced therapies poses a barrier to access for many patients.
- Regulatory Hurdles: Strict regulatory pathways can delay product launches and increase development costs.
- Competition: Intense competition among pharmaceutical companies influences pricing and market share.
- Late Diagnosis: Late diagnosis of ovarian cancer often leads to poorer outcomes and increases treatment costs.
Emerging Opportunities in Europe Ovarian Cancer Diagnostics and Therapeutics Industry
The European ovarian cancer market presents numerous opportunities for growth:
- Personalized Medicine: Tailoring treatment to individual patients' genetic profiles offers improved efficacy and reduced side effects.
- Biomarker Discovery: Identifying new biomarkers for early detection and treatment monitoring holds significant potential.
- Innovative Therapeutic Approaches: Emerging therapies, such as CAR T-cell therapy, offer promising avenues for treatment.
- Expansion into Emerging Markets: Untapped markets in Eastern Europe present opportunities for growth.
Leading Players in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry Market
Key Developments in Europe Ovarian Cancer Diagnostics and Therapeutics Industry Industry
August 2022: Inceptua Group commercially launched Apealea in Germany for the treatment of adult patients with the first relapse of platinum-sensitive epithelial ovarian cancer. This launch significantly expands treatment options for this patient population.
May 2022: BioMoti Ltd partnered with a global pharmaceutical company to collaborate on the development of its lead ovarian cancer candidate, BMT101, to clinical phase 2a proof-of-concept. This collaboration accelerates the development pipeline and could lead to new treatment options.
Future Outlook for Europe Ovarian Cancer Diagnostics and Therapeutics Industry Market
The future of the European ovarian cancer diagnostics and therapeutics market is bright, driven by continuous innovation in treatment modalities, growing awareness, and increased investments in research and development. The market is expected to witness significant growth, propelled by the adoption of personalized medicine approaches, the development of novel therapies, and expanded access to advanced diagnostics. Strategic collaborations and acquisitions will further shape the competitive landscape, leading to a dynamic and rapidly evolving market with substantial growth potential.
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Segmentation
-
1. Cancer Type
- 1.1. Epithelial Ovarian Tumors
- 1.2. Ovarian Germ Cell Tumors
- 1.3. Other Cancer Types
-
2. Modality
-
2.1. Diagnosis
- 2.1.1. Biopsy
- 2.1.2. Blood Tests
- 2.1.3. Ultrasound
- 2.1.4. PET
- 2.1.5. CT Scan
- 2.1.6. Other Diagnosis
-
2.2. Therapeutics
- 2.2.1. Chemotherapy
- 2.2.2. Radiation Therapy
- 2.2.3. Immunotherapy
- 2.2.4. Hormonal Therapy
- 2.2.5. Other Therapeutics
-
2.1. Diagnosis
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
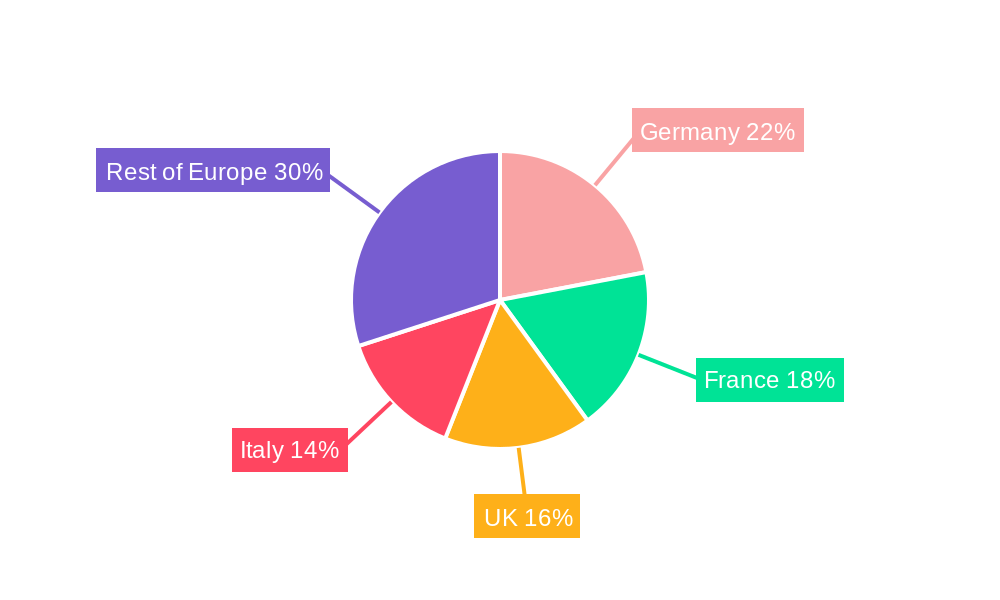
Europe Ovarian Cancer Diagnostics and Therapeutics Industry Regional Market Share

Geographic Coverage of Europe Ovarian Cancer Diagnostics and Therapeutics Industry
Europe Ovarian Cancer Diagnostics and Therapeutics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 14.99% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Cancer Type
- 5.1.1. Epithelial Ovarian Tumors
- 5.1.2. Ovarian Germ Cell Tumors
- 5.1.3. Other Cancer Types
- 5.2. Market Analysis, Insights and Forecast - by Modality
- 5.2.1. Diagnosis
- 5.2.1.1. Biopsy
- 5.2.1.2. Blood Tests
- 5.2.1.3. Ultrasound
- 5.2.1.4. PET
- 5.2.1.5. CT Scan
- 5.2.1.6. Other Diagnosis
- 5.2.2. Therapeutics
- 5.2.2.1. Chemotherapy
- 5.2.2.2. Radiation Therapy
- 5.2.2.3. Immunotherapy
- 5.2.2.4. Hormonal Therapy
- 5.2.2.5. Other Therapeutics
- 5.2.1. Diagnosis
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. Germany
- 5.3.2. United Kingdom
- 5.3.3. France
- 5.3.4. Italy
- 5.3.5. Spain
- 5.3.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Cancer Type
- 6. Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Cancer Type
- 6.1.1. Epithelial Ovarian Tumors
- 6.1.2. Ovarian Germ Cell Tumors
- 6.1.3. Other Cancer Types
- 6.2. Market Analysis, Insights and Forecast - by Modality
- 6.2.1. Diagnosis
- 6.2.1.1. Biopsy
- 6.2.1.2. Blood Tests
- 6.2.1.3. Ultrasound
- 6.2.1.4. PET
- 6.2.1.5. CT Scan
- 6.2.1.6. Other Diagnosis
- 6.2.2. Therapeutics
- 6.2.2.1. Chemotherapy
- 6.2.2.2. Radiation Therapy
- 6.2.2.3. Immunotherapy
- 6.2.2.4. Hormonal Therapy
- 6.2.2.5. Other Therapeutics
- 6.2.1. Diagnosis
- 6.1. Market Analysis, Insights and Forecast - by Cancer Type
- 7. Germany Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Cancer Type
- 7.1.1. Epithelial Ovarian Tumors
- 7.1.2. Ovarian Germ Cell Tumors
- 7.1.3. Other Cancer Types
- 7.2. Market Analysis, Insights and Forecast - by Modality
- 7.2.1. Diagnosis
- 7.2.1.1. Biopsy
- 7.2.1.2. Blood Tests
- 7.2.1.3. Ultrasound
- 7.2.1.4. PET
- 7.2.1.5. CT Scan
- 7.2.1.6. Other Diagnosis
- 7.2.2. Therapeutics
- 7.2.2.1. Chemotherapy
- 7.2.2.2. Radiation Therapy
- 7.2.2.3. Immunotherapy
- 7.2.2.4. Hormonal Therapy
- 7.2.2.5. Other Therapeutics
- 7.2.1. Diagnosis
- 7.1. Market Analysis, Insights and Forecast - by Cancer Type
- 8. United Kingdom Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Cancer Type
- 8.1.1. Epithelial Ovarian Tumors
- 8.1.2. Ovarian Germ Cell Tumors
- 8.1.3. Other Cancer Types
- 8.2. Market Analysis, Insights and Forecast - by Modality
- 8.2.1. Diagnosis
- 8.2.1.1. Biopsy
- 8.2.1.2. Blood Tests
- 8.2.1.3. Ultrasound
- 8.2.1.4. PET
- 8.2.1.5. CT Scan
- 8.2.1.6. Other Diagnosis
- 8.2.2. Therapeutics
- 8.2.2.1. Chemotherapy
- 8.2.2.2. Radiation Therapy
- 8.2.2.3. Immunotherapy
- 8.2.2.4. Hormonal Therapy
- 8.2.2.5. Other Therapeutics
- 8.2.1. Diagnosis
- 8.1. Market Analysis, Insights and Forecast - by Cancer Type
- 9. France Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Cancer Type
- 9.1.1. Epithelial Ovarian Tumors
- 9.1.2. Ovarian Germ Cell Tumors
- 9.1.3. Other Cancer Types
- 9.2. Market Analysis, Insights and Forecast - by Modality
- 9.2.1. Diagnosis
- 9.2.1.1. Biopsy
- 9.2.1.2. Blood Tests
- 9.2.1.3. Ultrasound
- 9.2.1.4. PET
- 9.2.1.5. CT Scan
- 9.2.1.6. Other Diagnosis
- 9.2.2. Therapeutics
- 9.2.2.1. Chemotherapy
- 9.2.2.2. Radiation Therapy
- 9.2.2.3. Immunotherapy
- 9.2.2.4. Hormonal Therapy
- 9.2.2.5. Other Therapeutics
- 9.2.1. Diagnosis
- 9.1. Market Analysis, Insights and Forecast - by Cancer Type
- 10. Italy Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Cancer Type
- 10.1.1. Epithelial Ovarian Tumors
- 10.1.2. Ovarian Germ Cell Tumors
- 10.1.3. Other Cancer Types
- 10.2. Market Analysis, Insights and Forecast - by Modality
- 10.2.1. Diagnosis
- 10.2.1.1. Biopsy
- 10.2.1.2. Blood Tests
- 10.2.1.3. Ultrasound
- 10.2.1.4. PET
- 10.2.1.5. CT Scan
- 10.2.1.6. Other Diagnosis
- 10.2.2. Therapeutics
- 10.2.2.1. Chemotherapy
- 10.2.2.2. Radiation Therapy
- 10.2.2.3. Immunotherapy
- 10.2.2.4. Hormonal Therapy
- 10.2.2.5. Other Therapeutics
- 10.2.1. Diagnosis
- 10.1. Market Analysis, Insights and Forecast - by Cancer Type
- 11. Spain Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Cancer Type
- 11.1.1. Epithelial Ovarian Tumors
- 11.1.2. Ovarian Germ Cell Tumors
- 11.1.3. Other Cancer Types
- 11.2. Market Analysis, Insights and Forecast - by Modality
- 11.2.1. Diagnosis
- 11.2.1.1. Biopsy
- 11.2.1.2. Blood Tests
- 11.2.1.3. Ultrasound
- 11.2.1.4. PET
- 11.2.1.5. CT Scan
- 11.2.1.6. Other Diagnosis
- 11.2.2. Therapeutics
- 11.2.2.1. Chemotherapy
- 11.2.2.2. Radiation Therapy
- 11.2.2.3. Immunotherapy
- 11.2.2.4. Hormonal Therapy
- 11.2.2.5. Other Therapeutics
- 11.2.1. Diagnosis
- 11.1. Market Analysis, Insights and Forecast - by Cancer Type
- 12. Rest of Europe Europe Ovarian Cancer Diagnostics and Therapeutics Industry Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Cancer Type
- 12.1.1. Epithelial Ovarian Tumors
- 12.1.2. Ovarian Germ Cell Tumors
- 12.1.3. Other Cancer Types
- 12.2. Market Analysis, Insights and Forecast - by Modality
- 12.2.1. Diagnosis
- 12.2.1.1. Biopsy
- 12.2.1.2. Blood Tests
- 12.2.1.3. Ultrasound
- 12.2.1.4. PET
- 12.2.1.5. CT Scan
- 12.2.1.6. Other Diagnosis
- 12.2.2. Therapeutics
- 12.2.2.1. Chemotherapy
- 12.2.2.2. Radiation Therapy
- 12.2.2.3. Immunotherapy
- 12.2.2.4. Hormonal Therapy
- 12.2.2.5. Other Therapeutics
- 12.2.1. Diagnosis
- 12.1. Market Analysis, Insights and Forecast - by Cancer Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 INEX Innovate
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Boehringer Ingelheim International GmbH
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 AstraZeneca PLC
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Eli Lilly and Company
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Siemens Healthineers AG
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Johnson and Johnson (Janssen Pharmaceuticals)
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Ovation Diagnostics
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Bristol Myers Squibb Company
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 F Hoffman-La Roche Ltd
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Amneal Pharmaceuticals LLC
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.11 GlaxoSmithKline PLC
- 13.1.11.1. Company Overview
- 13.1.11.2. Products
- 13.1.11.3. Company Financials
- 13.1.11.4. SWOT Analysis
- 13.1.12 Pfizer Inc
- 13.1.12.1. Company Overview
- 13.1.12.2. Products
- 13.1.12.3. Company Financials
- 13.1.12.4. SWOT Analysis
- 13.1.1 INEX Innovate
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Share (%) by Company 2025
List of Tables
- Table 1: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 2: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 3: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 4: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 5: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 8: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 9: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 10: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 11: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 14: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 15: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 16: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 17: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 18: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 19: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 20: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 21: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 22: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 23: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 26: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 27: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 28: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 29: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 30: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 31: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 32: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 33: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 34: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 35: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 36: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 37: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Cancer Type 2020 & 2033
- Table 38: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Cancer Type 2020 & 2033
- Table 39: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Modality 2020 & 2033
- Table 40: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Modality 2020 & 2033
- Table 41: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 42: Europe Ovarian Cancer Diagnostics and Therapeutics Industry Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Ovarian Cancer Diagnostics and Therapeutics Industry?
The projected CAGR is approximately 14.99%.
2. Which companies are prominent players in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry?
Key companies in the market include INEX Innovate, Boehringer Ingelheim International GmbH, AstraZeneca PLC, Eli Lilly and Company, Siemens Healthineers AG, Johnson and Johnson (Janssen Pharmaceuticals), Ovation Diagnostics, Bristol Myers Squibb Company, F Hoffman-La Roche Ltd, Amneal Pharmaceuticals LLC, GlaxoSmithKline PLC, Pfizer Inc.
3. What are the main segments of the Europe Ovarian Cancer Diagnostics and Therapeutics Industry?
The market segments include Cancer Type, Modality.
4. Can you provide details about the market size?
The market size is estimated to be USD 7.62 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Burden of Ovarian Cancer; Greater Use of Combination Therapies for the Treatment of Ovarian Cancer; Rising Geriatric Population.
6. What are the notable trends driving market growth?
Immunotherapy is Expected to Hold Significant Share of the European Ovarian Cancer Diagnostics and Therapeutics Market During the Forecast Period.
7. Are there any restraints impacting market growth?
Lack of Accurate Diagnosis of Ovarian Cancer.
8. Can you provide examples of recent developments in the market?
August 2022: Inceptua Group commercially launched Apealea in Germany for the treatment of adult patients with the first relapse of platinum-sensitive epithelial ovarian cancer.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Ovarian Cancer Diagnostics and Therapeutics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry?
To stay informed about further developments, trends, and reports in the Europe Ovarian Cancer Diagnostics and Therapeutics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
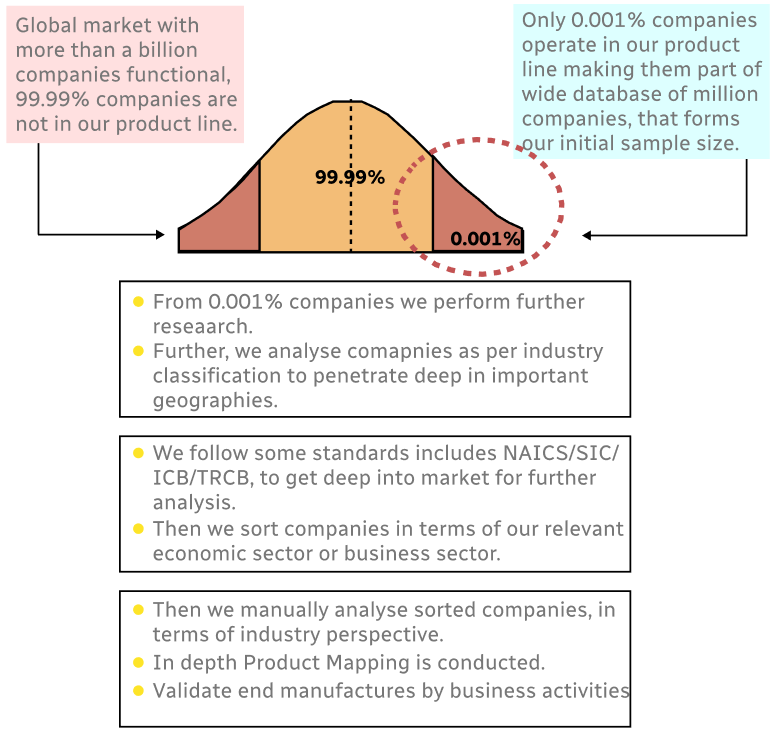
Step 1 - Identification of Relevant Samples Size from Population Database
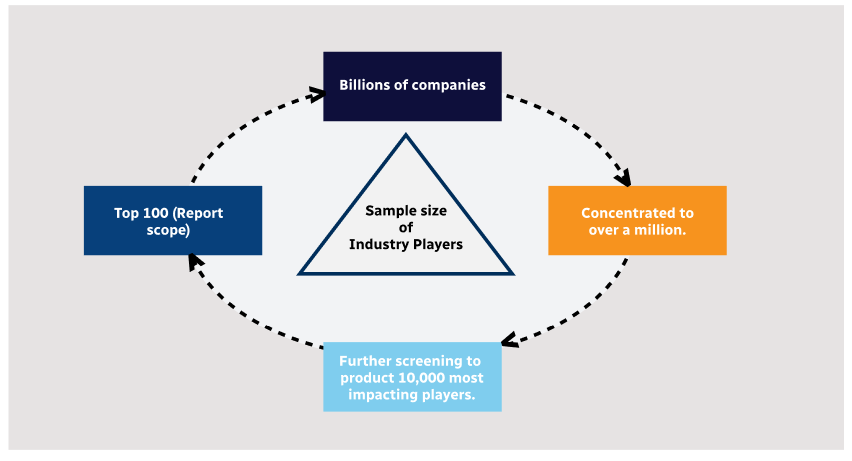
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
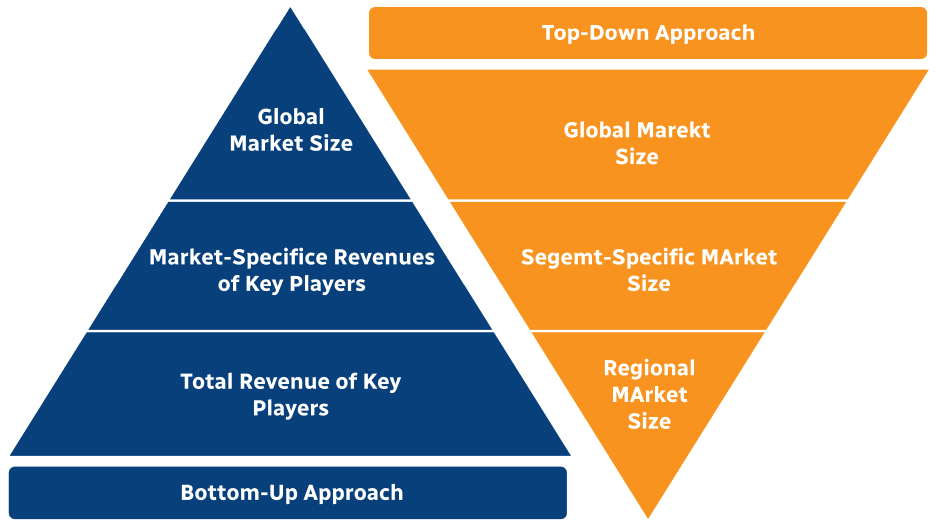
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


