Key Insights
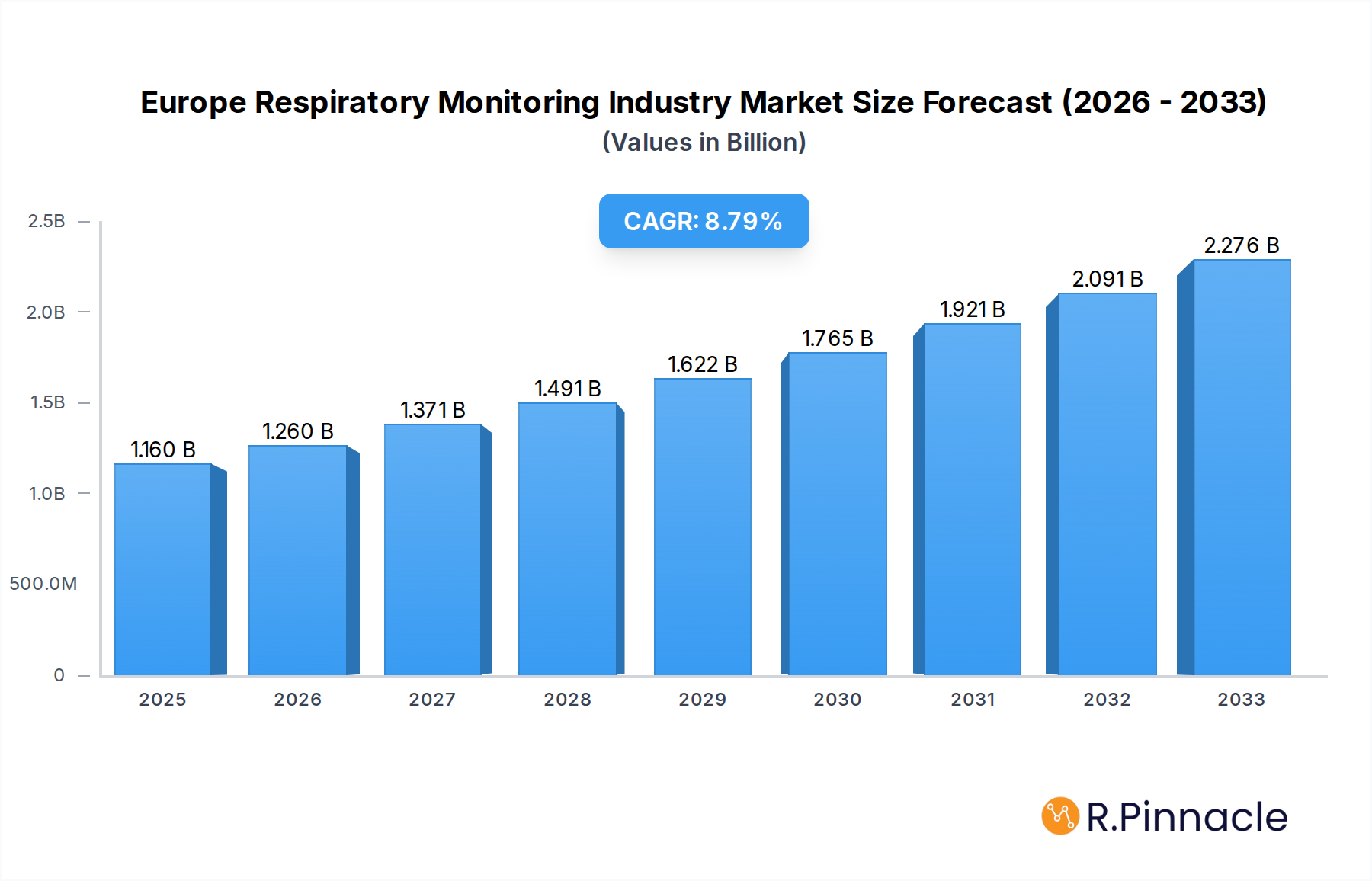
The Europe Respiratory Monitoring Industry is poised for significant expansion, projected to reach $1.16 billion by 2025, with a compelling Compound Annual Growth Rate (CAGR) of 8.60% anticipated to propel it through to 2033. This robust growth is underpinned by a confluence of factors, including the increasing prevalence of respiratory diseases such as asthma, COPD, and sleep apnea across the continent, driven by aging populations, rising pollution levels, and sedentary lifestyles. Furthermore, advancements in technology, leading to the development of more accurate, portable, and user-friendly respiratory monitoring devices, are significantly contributing to market penetration. The growing adoption of home-based patient monitoring solutions, fueled by a desire for convenience and cost-effectiveness, alongside supportive government initiatives promoting respiratory health awareness and early diagnosis, further acts as a powerful catalyst for market expansion.
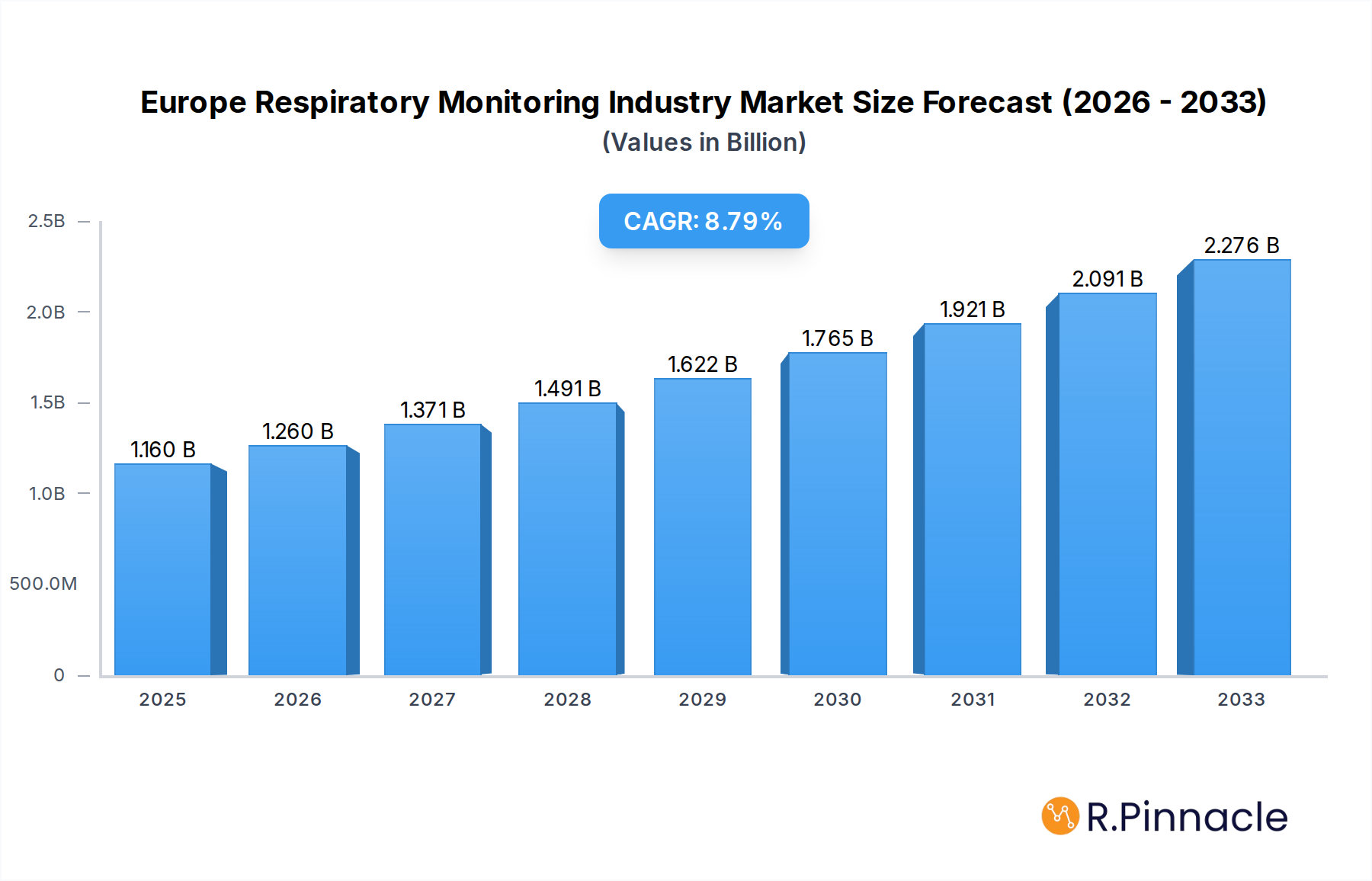
Europe Respiratory Monitoring Industry Market Size (In Billion)

The market is segmented across a diverse range of device types, with Spirometers and Pulse Oximeters holding substantial market share due to their widespread application in diagnosis and ongoing management of respiratory conditions. Sleep Test Devices are also witnessing a surge in demand driven by increased awareness and diagnosis of sleep-related breathing disorders. Leading companies like Maquet (Getinge AB), Vitalograph Ltd, Medtronic, GE Healthcare, and Koninklijke Philips N.V. are at the forefront, investing heavily in research and development to innovate and capture market share. While the market is characterized by strong growth drivers, potential restraints include stringent regulatory approvals for new medical devices and the high initial cost of some advanced monitoring systems, though the increasing focus on value-based healthcare and reimbursement policies are expected to mitigate these challenges over the forecast period. The European region, with its well-established healthcare infrastructure and high disposable incomes, presents a fertile ground for the sustained growth of the respiratory monitoring industry.
Europe Respiratory Monitoring Industry Company Market Share

This in-depth report provides a definitive analysis of the Europe Respiratory Monitoring Industry, offering critical insights into market size, growth drivers, competitive landscape, and future projections. Spanning the historical period of 2019-2024, the base year of 2025, and a robust forecast period from 2025-2033, this report equips industry professionals with actionable intelligence to navigate this dynamic sector. Discover key trends shaping respiratory care, from the proliferation of advanced spirometers and portable pulse oximeters to the growing demand for sleep test devices and sophisticated gas analyzers. Our analysis delves into regulatory shifts, technological innovations, and emerging market opportunities, making this an indispensable resource for manufacturers, suppliers, healthcare providers, and investors.
Europe Respiratory Monitoring Industry Market Structure & Innovation Trends
The Europe Respiratory Monitoring Industry is characterized by a moderately concentrated market structure, with a few key players holding significant market share, estimated at approximately 60% for the top five entities. Innovation is a primary driver, fueled by advancements in sensor technology, data analytics, and connectivity, enabling real-time patient monitoring and remote diagnostics. Regulatory frameworks, such as those governed by the European Medicines Agency (EMA) and national health authorities, play a crucial role in ensuring product safety and efficacy, influencing market entry and product development strategies. Product substitutes, while present in the form of traditional diagnostic methods, are increasingly being superseded by integrated digital solutions. End-user demographics span a broad spectrum, from aging populations with a higher prevalence of respiratory conditions to athletes and occupational health settings. Mergers and acquisitions (M&A) activities are prevalent, with recent deal values estimated to be in the hundreds of millions of Euros, as companies strategically consolidate to enhance their product portfolios and market reach. Key areas of focus for innovation include miniaturization of devices, AI-powered diagnostic assistance, and user-friendly interfaces for both clinical and home-use applications.
Europe Respiratory Monitoring Industry Market Dynamics & Trends
The Europe Respiratory Monitoring Industry is poised for significant expansion, driven by a confluence of compelling market growth drivers, disruptive technological advancements, evolving consumer preferences, and intense competitive dynamics. A primary catalyst for this growth is the escalating prevalence of chronic respiratory diseases, such as Chronic Obstructive Pulmonary Disease (COPD) and asthma, across the European continent. This demographic shift necessitates a greater demand for accurate, reliable, and accessible respiratory monitoring solutions. Furthermore, the ongoing global healthcare paradigm shift towards proactive and preventative care, coupled with a growing emphasis on home-based patient management, is significantly boosting the adoption of portable and connected respiratory monitoring devices. Technological disruptions are at the forefront of market evolution. The integration of Artificial Intelligence (AI) and Machine Learning (ML) into respiratory monitoring devices is enabling sophisticated data analysis, predictive diagnostics, and personalized treatment plans. Wireless connectivity, including Bluetooth and Wi-Fi, facilitates seamless data transfer to healthcare professionals and electronic health records (EHRs), enhancing remote patient monitoring capabilities and reducing the burden on healthcare infrastructure. Consumer preferences are increasingly leaning towards user-friendly, non-invasive, and smart devices that empower individuals to actively manage their respiratory health. This includes a rising demand for wearable respiratory trackers and mobile spirometry applications. The competitive landscape is dynamic, with established players constantly innovating and smaller, agile companies emerging with niche solutions. Strategic partnerships and collaborations are becoming crucial for market players to expand their geographical reach and technological capabilities. Market penetration for advanced respiratory monitoring solutions is projected to grow from approximately 35% in the base year 2025 to an estimated 55% by 2033, indicating substantial room for continued growth. The Compound Annual Growth Rate (CAGR) for the Europe Respiratory Monitoring Industry is forecast to be in the range of 7.5% to 9.0% during the forecast period of 2025–2033, underscoring a robust upward trajectory.
Dominant Regions & Segments in Europe Respiratory Monitoring Industry
The Germany stands as the dominant region within the Europe Respiratory Monitoring Industry, driven by its robust healthcare infrastructure, high disposable income, and a proactive approach to adopting advanced medical technologies. Significant government investment in healthcare, coupled with a strong emphasis on preventative medicine and chronic disease management, further solidifies Germany's leading position. The country's advanced research and development capabilities also contribute to its prominence in driving innovation within the sector.
Within the Type of Device segmentation, Spirometers currently hold the largest market share, estimated at over 30% of the total market value. This dominance is attributed to their foundational role in diagnosing and monitoring a wide range of respiratory conditions, including asthma and COPD. Their widespread use in pulmonary function testing (PFT) labs, hospitals, and increasingly in primary care settings underscores their essential nature.
Key Drivers for German Dominance:
- Advanced Healthcare Infrastructure: Extensive network of hospitals, specialized clinics, and research institutions.
- High Disposable Income: Enables greater affordability of advanced medical devices for both individuals and healthcare providers.
- Government Healthcare Policies: Strong support for R&D, medical device innovation, and public health initiatives.
- Technological Adoption: Germany is an early adopter of cutting-edge medical technologies and digital health solutions.
- Aging Population: A significant demographic segment prone to respiratory ailments, driving consistent demand.
Dominance of Spirometers:
- Diagnostic Cornerstone: Essential for diagnosing conditions like asthma, COPD, and cystic fibrosis.
- Therapeutic Monitoring: Crucial for assessing treatment efficacy and disease progression.
- Clinical Ubiquity: Standard equipment in pulmonology departments, general practices, and occupational health screenings.
- Technological Advancements: Integration of digital features, wireless connectivity, and user-friendly software enhances their appeal and utility.
The Sleep Test Devices segment is experiencing the fastest growth, projected to witness a CAGR of approximately 10.5% during the forecast period. This surge is fueled by the increasing recognition of sleep disorders' impact on overall health and the growing demand for home sleep apnea testing (HSAT) solutions.
Pulse Oximeters also represent a significant and growing segment, valued at an estimated 15% of the market. Their portability, non-invasiveness, and critical role in monitoring oxygen saturation levels make them indispensable for acute care, chronic disease management, and remote patient monitoring. The increasing prevalence of conditions like pneumonia and COVID-19 has further amplified their demand.
Gas Analyzers and Capnographs are specialized yet vital segments, catering to critical care settings, anesthesia, and intensive respiratory support. While smaller in market share, their high-value applications ensure consistent demand and technological innovation.
Europe Respiratory Monitoring Industry Product Innovations
Product innovations in the Europe Respiratory Monitoring Industry are centered on enhancing accuracy, portability, and user-friendliness. Companies are developing miniaturized spirometers and integrated diagnostic platforms that combine multiple monitoring functions in single devices. The integration of AI and machine learning is enabling predictive analytics for early detection of respiratory exacerbations and personalized treatment recommendations. Advancements in wireless connectivity and cloud-based platforms are facilitating seamless remote patient monitoring, allowing healthcare providers to track patient status in real-time, thereby improving patient outcomes and reducing hospital readmissions. These innovations offer distinct competitive advantages by providing more comprehensive data, improved patient adherence, and greater operational efficiency for healthcare systems.
Report Scope & Segmentation Analysis
This report comprehensively analyzes the Europe Respiratory Monitoring Industry across key segments. The Spirometers segment is projected to maintain its leading market position, driven by widespread clinical adoption and ongoing technological enhancements. Peak Flow Meters will continue to serve the needs of asthma management, particularly for home use, with steady growth anticipated. Sleep Test Devices are expected to exhibit the highest growth rate, fueled by increasing awareness of sleep disorders and the demand for convenient home testing solutions. Gas Analyzers will cater to specialized applications in critical care and anesthesia, with stable demand. Pulse Oximeters will experience robust growth due to their versatility in monitoring oxygen saturation for various respiratory conditions, both in clinical and home settings. The Others segment will encompass emerging technologies and niche devices with potential for future expansion.
Key Drivers of Europe Respiratory Monitoring Industry Growth
The Europe Respiratory Monitoring Industry is propelled by several key growth drivers. Technologically, the increasing sophistication of sensor technology, coupled with advancements in AI and connectivity, enables more accurate, real-time, and remote monitoring. Economically, the rising healthcare expenditure across Europe and the growing burden of chronic respiratory diseases necessitate more efficient and proactive patient management solutions. Regulatory support for digital health initiatives and telehealth further encourages the adoption of advanced monitoring devices. The increasing demand for home-based care and patient empowerment in managing their own health is also a significant driver, pushing the market towards portable and user-friendly devices.
Challenges in the Europe Respiratory Monitoring Industry Sector
Despite its growth, the Europe Respiratory Monitoring Industry faces several challenges. Stringent regulatory hurdles and the lengthy approval processes for new medical devices can impede market entry and innovation. Reimbursement policies for remote monitoring solutions and advanced diagnostic equipment can vary significantly across European countries, creating inconsistencies in market access. Supply chain disruptions, particularly those exacerbated by global events, can impact the availability and cost of essential components. Furthermore, the competitive pressure from both established global players and agile startups necessitates continuous innovation and cost-effective solutions. Data security and privacy concerns associated with connected devices also pose a significant challenge that needs to be meticulously addressed.
Emerging Opportunities in Europe Respiratory Monitoring Industry
Emerging opportunities within the Europe Respiratory Monitoring Industry lie in the burgeoning field of digital health and personalized medicine. The expansion of telehealth platforms presents a significant avenue for increased adoption of remote respiratory monitoring devices, enabling continuous patient care outside traditional clinical settings. The growing emphasis on preventative healthcare and early disease detection opens doors for more sophisticated screening tools and wearable sensors that can identify subtle changes in respiratory function. Furthermore, the development of AI-powered diagnostic algorithms and data analytics solutions offers substantial potential for improving diagnostic accuracy and treatment personalization. Untapped markets within Eastern Europe, with their growing healthcare investments, also present significant growth prospects for market players.
Leading Players in the Europe Respiratory Monitoring Industry Market
- Maquet (Getinge AB)
- Vitalograph Ltd
- Medtronic
- GE Healthcare
- Koninklijke Philips N V
- COSMED srl
- Resmed
- Siemens Healthcare GmbH
- Koko PFT
- Draegerwerk AG
Key Developments in Europe Respiratory Monitoring Industry Industry
- March 2022: KoKo, LLC partnered with MAGNET GROUP, a three-year agreement allowing MAGNET GROUP to leverage KoKo's high-quality spirometry diagnostics and software.
- March 2021: Vyaire Medical launched its AioCare mobile spirometry system in over 15 countries across Europe, Australia, and the Middle East.
Future Outlook for Europe Respiratory Monitoring Industry Market
The future outlook for the Europe Respiratory Monitoring Industry is exceptionally bright, driven by sustained demand for advanced respiratory care solutions and accelerating technological integration. The increasing adoption of telehealth and remote patient monitoring will continue to fuel the market for connected and user-friendly devices. Innovations in AI and data analytics will lead to more predictive and personalized respiratory management strategies, enhancing patient outcomes. The aging European population and the persistent prevalence of respiratory diseases will ensure a robust underlying demand. Strategic partnerships and collaborations will remain crucial for market players to expand their reach and technological capabilities. The market is poised for significant growth, with substantial opportunities for companies that can offer innovative, cost-effective, and compliant respiratory monitoring solutions.
Europe Respiratory Monitoring Industry Segmentation
-
1. Type of Device
- 1.1. Spirometers
- 1.2. Peak Flow Meters
- 1.3. Sleep Test Devices
- 1.4. Gas Analyzers
- 1.5. Pulse Oximeters
- 1.6. Capnographs
- 1.7. Others
Europe Respiratory Monitoring Industry Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
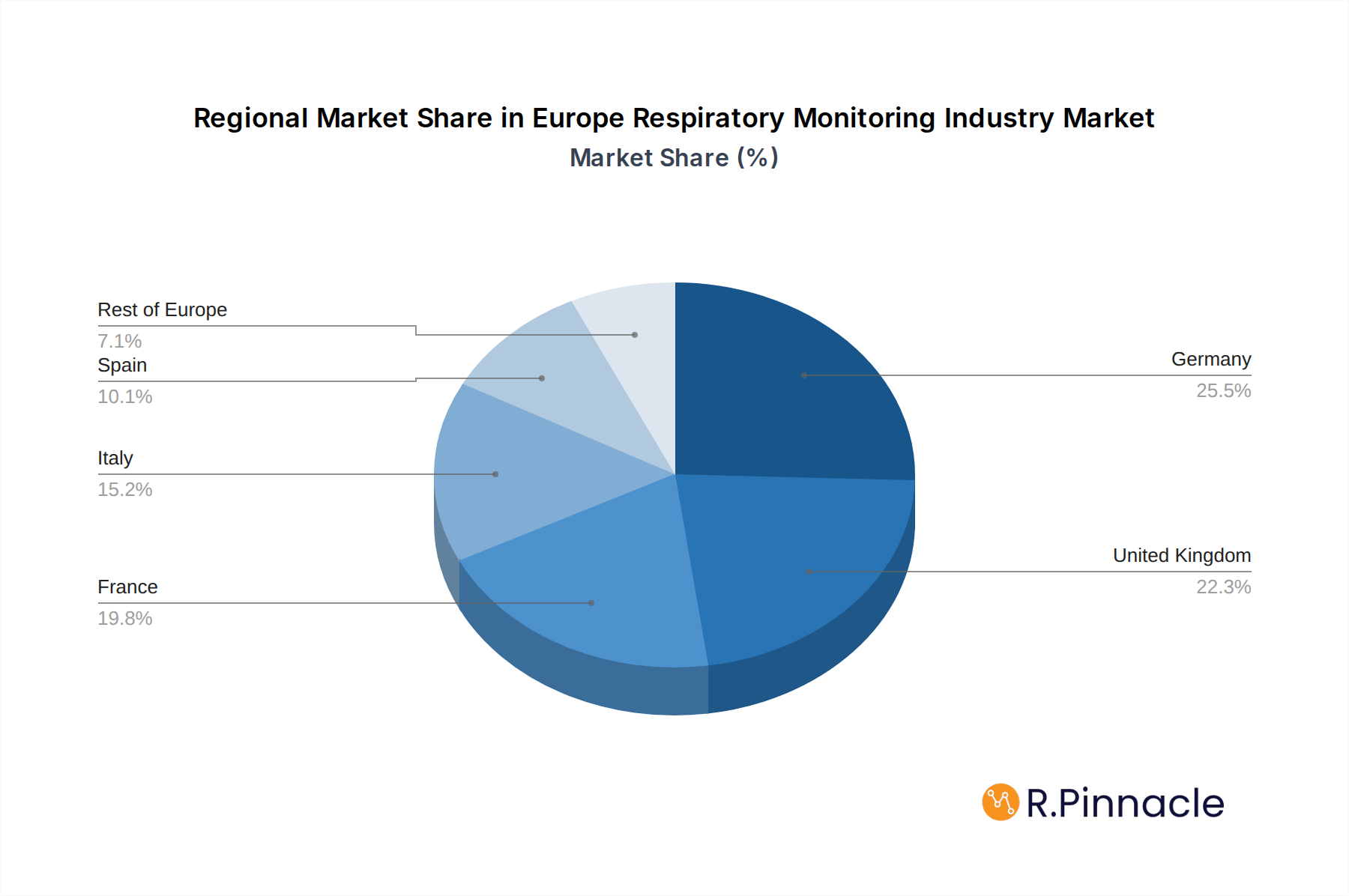
Europe Respiratory Monitoring Industry Regional Market Share

Geographic Coverage of Europe Respiratory Monitoring Industry
Europe Respiratory Monitoring Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.60% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 5.1.1. Spirometers
- 5.1.2. Peak Flow Meters
- 5.1.3. Sleep Test Devices
- 5.1.4. Gas Analyzers
- 5.1.5. Pulse Oximeters
- 5.1.6. Capnographs
- 5.1.7. Others
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Germany
- 5.2.2. United Kingdom
- 5.2.3. France
- 5.2.4. Italy
- 5.2.5. Spain
- 5.2.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 6. Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 6.1.1. Spirometers
- 6.1.2. Peak Flow Meters
- 6.1.3. Sleep Test Devices
- 6.1.4. Gas Analyzers
- 6.1.5. Pulse Oximeters
- 6.1.6. Capnographs
- 6.1.7. Others
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 7. Germany Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Type of Device
- 7.1.1. Spirometers
- 7.1.2. Peak Flow Meters
- 7.1.3. Sleep Test Devices
- 7.1.4. Gas Analyzers
- 7.1.5. Pulse Oximeters
- 7.1.6. Capnographs
- 7.1.7. Others
- 7.1. Market Analysis, Insights and Forecast - by Type of Device
- 8. United Kingdom Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Type of Device
- 8.1.1. Spirometers
- 8.1.2. Peak Flow Meters
- 8.1.3. Sleep Test Devices
- 8.1.4. Gas Analyzers
- 8.1.5. Pulse Oximeters
- 8.1.6. Capnographs
- 8.1.7. Others
- 8.1. Market Analysis, Insights and Forecast - by Type of Device
- 9. France Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Type of Device
- 9.1.1. Spirometers
- 9.1.2. Peak Flow Meters
- 9.1.3. Sleep Test Devices
- 9.1.4. Gas Analyzers
- 9.1.5. Pulse Oximeters
- 9.1.6. Capnographs
- 9.1.7. Others
- 9.1. Market Analysis, Insights and Forecast - by Type of Device
- 10. Italy Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Type of Device
- 10.1.1. Spirometers
- 10.1.2. Peak Flow Meters
- 10.1.3. Sleep Test Devices
- 10.1.4. Gas Analyzers
- 10.1.5. Pulse Oximeters
- 10.1.6. Capnographs
- 10.1.7. Others
- 10.1. Market Analysis, Insights and Forecast - by Type of Device
- 11. Spain Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Type of Device
- 11.1.1. Spirometers
- 11.1.2. Peak Flow Meters
- 11.1.3. Sleep Test Devices
- 11.1.4. Gas Analyzers
- 11.1.5. Pulse Oximeters
- 11.1.6. Capnographs
- 11.1.7. Others
- 11.1. Market Analysis, Insights and Forecast - by Type of Device
- 12. Rest of Europe Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Type of Device
- 12.1.1. Spirometers
- 12.1.2. Peak Flow Meters
- 12.1.3. Sleep Test Devices
- 12.1.4. Gas Analyzers
- 12.1.5. Pulse Oximeters
- 12.1.6. Capnographs
- 12.1.7. Others
- 12.1. Market Analysis, Insights and Forecast - by Type of Device
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Maquet (Getinge AB)
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Vitalograph Ltd
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Medtronic
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 GE Healthcare
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Koninklijke Philips N V
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 COSMED srl
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Resmed
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Siemens Healthcare GmbH
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Koko PFT
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Draegerwerk AG
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.1 Maquet (Getinge AB)
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Respiratory Monitoring Industry Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: Europe Respiratory Monitoring Industry Share (%) by Company 2025
List of Tables
- Table 1: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 2: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 3: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 4: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 5: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 6: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 7: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 8: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 10: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 11: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 12: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 13: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 14: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Respiratory Monitoring Industry?
The projected CAGR is approximately 8.60%.
2. Which companies are prominent players in the Europe Respiratory Monitoring Industry?
Key companies in the market include Maquet (Getinge AB), Vitalograph Ltd, Medtronic, GE Healthcare, Koninklijke Philips N V, COSMED srl, Resmed, Siemens Healthcare GmbH, Koko PFT, Draegerwerk AG.
3. What are the main segments of the Europe Respiratory Monitoring Industry?
The market segments include Type of Device.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.16 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Respiratory Diseases; Technological Advancements; Growing Geriatric Population.
6. What are the notable trends driving market growth?
Peak Flow Meters are Expected to Register Significant Growth over the Forecast Period.
7. Are there any restraints impacting market growth?
Reimbursement Concerns; High Cost Associated with the Devices.
8. Can you provide examples of recent developments in the market?
In March 2022, KoKo, LLC, a United Kingdom-based manufacturer of spirometers, signed a three-year partnership with MAGNET GROUP. This partnership would allow MAGNET GROUP to take advantage of the high-quality testing capabilities and features of KoKo's lung function diagnostics and software for spirometry and other devices.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Respiratory Monitoring Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Respiratory Monitoring Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Respiratory Monitoring Industry?
To stay informed about further developments, trends, and reports in the Europe Respiratory Monitoring Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
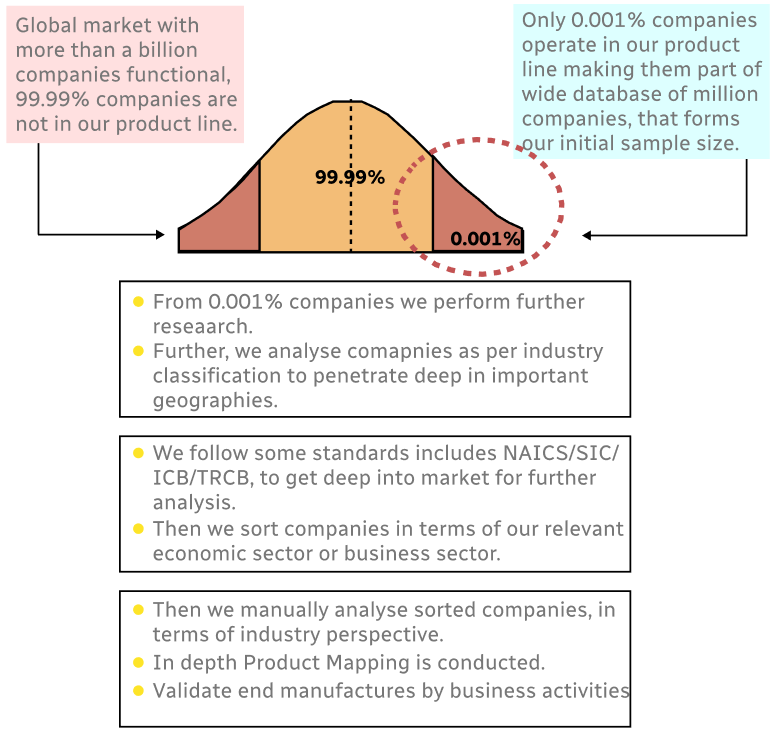
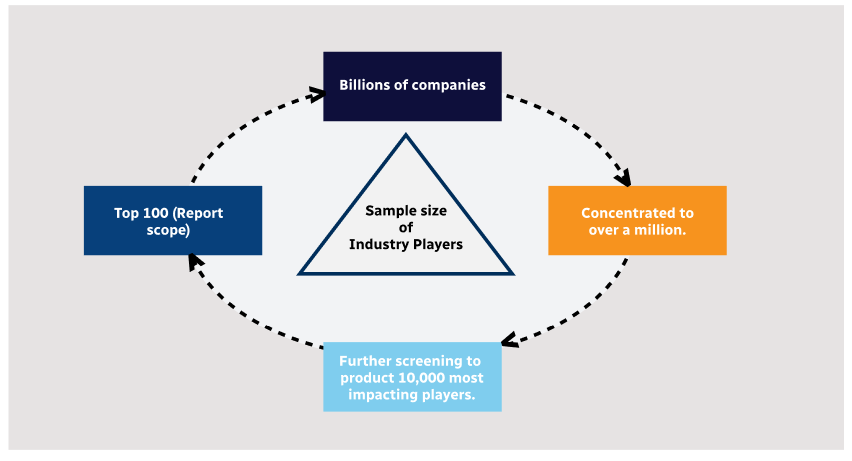
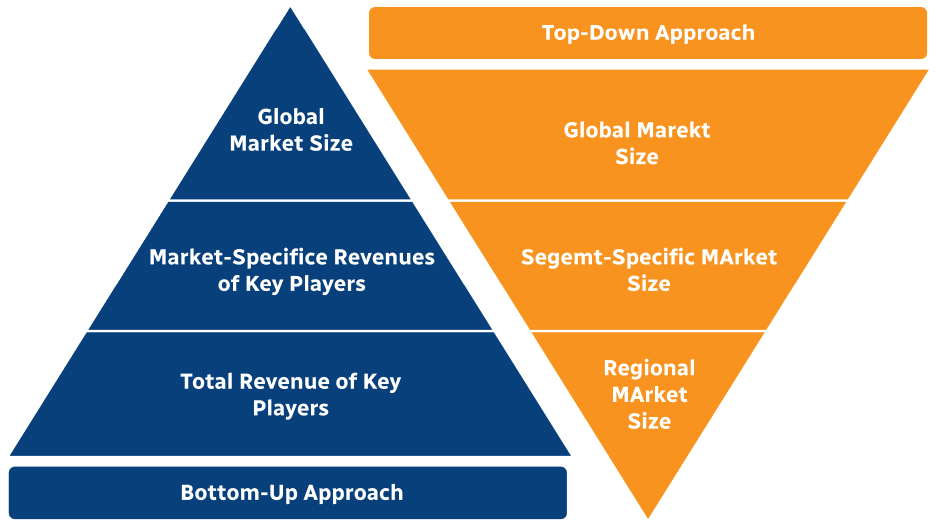
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
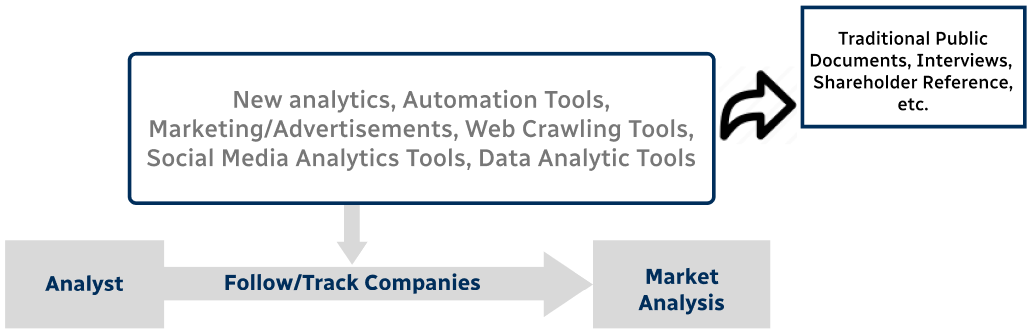
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


