Key Insights
The global market for Integrated Biomedical Testing Equipment is poised for significant expansion, projected to reach an estimated $171 million by 2025, with a robust Compound Annual Growth Rate (CAGR) of 4.6% from 2019 to 2033. This growth trajectory is fueled by escalating demand for advanced medical devices and the imperative for stringent quality control and patient safety in healthcare settings. The increasing prevalence of chronic diseases, a growing elderly population, and continuous technological advancements in medical equipment are key drivers pushing the adoption of these sophisticated testing solutions. Moreover, stringent regulatory requirements and the need for accurate calibration and maintenance of complex biomedical instruments further bolster market expansion. The market is segmented by application into Hospitals, Clinics, and Others, with hospitals representing the largest segment due to their extensive use of diverse medical equipment.
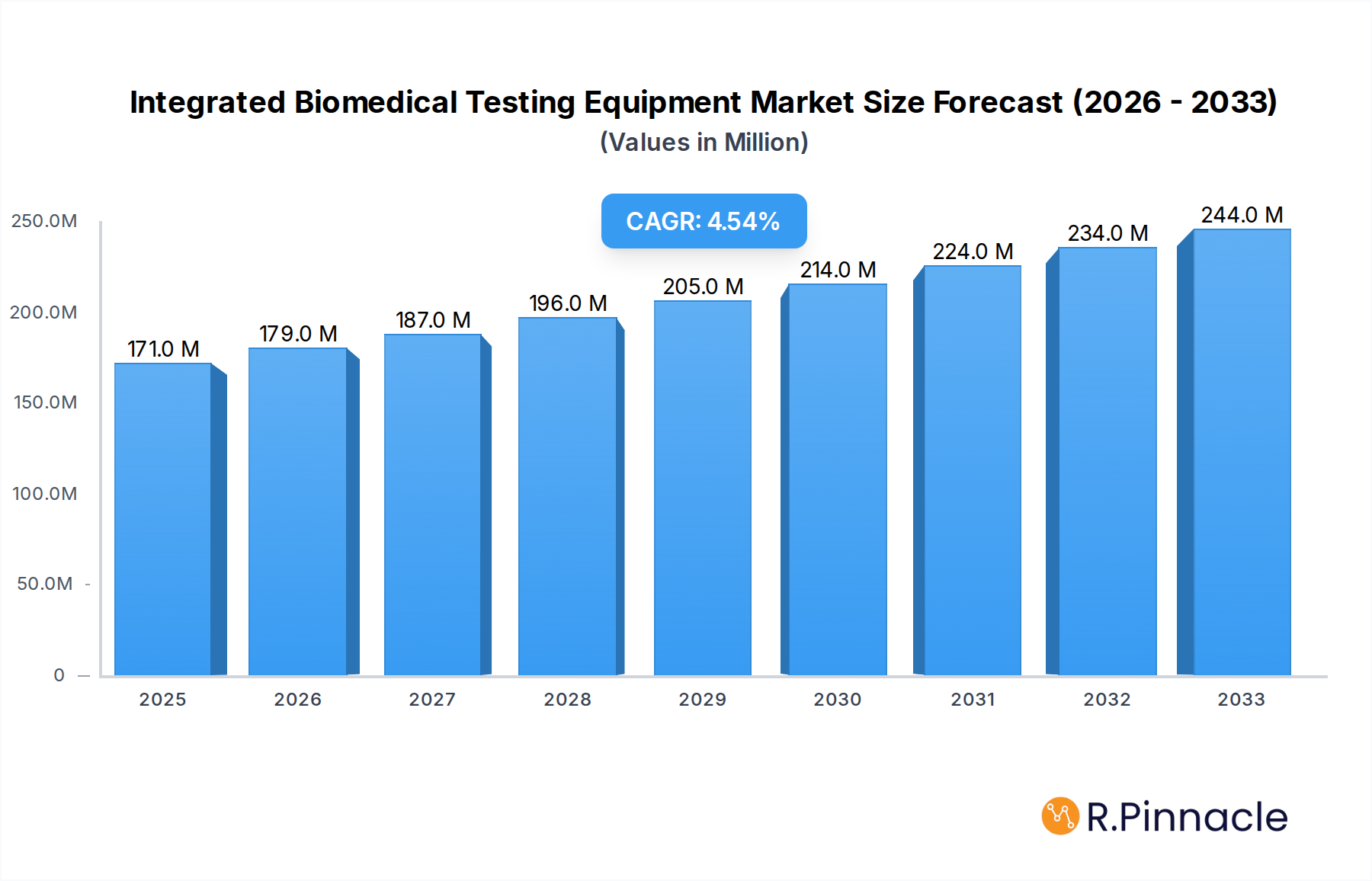
Integrated Biomedical Testing Equipment Market Size (In Million)

The Integrated Biomedical Testing Equipment market encompasses a wide array of product types, including Infusion Pump Analyzers, Defibrillator Analyzers, Electrosurgery Analyzers, Gas Flow Analyzers, Vital Sign Simulators, and Electrical Safety Analyzers. The growing complexity and sophistication of these medical devices necessitate specialized testing equipment for their accurate performance verification and safety assurance. Key market players, including Fluke Corporation, Gossen Metrawatt GmbH, and BC Group International, are actively engaged in research and development to introduce innovative solutions, enhancing the capabilities and efficiency of biomedical testing. Geographically, North America and Europe currently dominate the market, driven by their well-established healthcare infrastructures and high adoption rates of advanced medical technologies. However, the Asia Pacific region is expected to witness the fastest growth, propelled by rapid advancements in healthcare facilities and increasing investments in medical device manufacturing and maintenance in countries like China and India. Challenges, such as the high cost of advanced testing equipment and the need for skilled personnel, may slightly temper growth but are unlikely to impede the overall positive outlook.
Integrated Biomedical Testing Equipment Company Market Share

Here is the SEO-optimized, reader-centric report description for Integrated Biomedical Testing Equipment, designed for immediate use without modification.
Integrated Biomedical Testing Equipment Market Structure & Innovation Trends
The integrated biomedical testing equipment market exhibits a moderately concentrated structure, with key players like Fluke Corporation, Gossen Metrawatt GmbH, and BC Group International holding significant market share, estimated to be over 25% collectively by the base year 2025. Innovation is primarily driven by the increasing demand for advanced diagnostic accuracy, the integration of IoT and AI in medical devices, and stringent regulatory compliance requirements. This has led to a surge in the development of multi-functional analyzers capable of performing a wide array of tests. Regulatory frameworks, such as FDA guidelines and CE marking, play a crucial role in shaping product development and market entry, ensuring patient safety and device efficacy. Product substitutes, while present in the form of standalone testing devices, are increasingly being overshadowed by integrated solutions offering greater efficiency and cost-effectiveness. End-user demographics are shifting towards larger hospital networks and expanding healthcare facilities in emerging economies, demanding robust and scalable testing solutions. Mergers and acquisitions (M&A) activity is expected to remain steady, with an estimated total deal value exceeding $500 million during the forecast period, as larger companies seek to acquire innovative technologies and expand their product portfolios to consolidate their market position. Key M&A targets are likely to be smaller, specialized technology firms focusing on niche testing segments or advanced software integration.
Integrated Biomedical Testing Equipment Market Dynamics & Trends
The integrated biomedical testing equipment market is poised for robust growth, driven by an escalating global demand for advanced healthcare infrastructure and stringent medical device safety standards. The increasing prevalence of chronic diseases and an aging global population necessitate sophisticated diagnostic and maintenance tools for a vast array of medical equipment, fueling the adoption of these specialized testing solutions. Technological advancements are at the forefront of market evolution, with a pronounced trend towards miniaturization, enhanced automation, and the integration of artificial intelligence (AI) and the Internet of Medical Things (IoMT) into testing equipment. These innovations enable real-time data analysis, predictive maintenance, and remote monitoring capabilities, significantly improving operational efficiency and reducing downtime in healthcare facilities. The market penetration of integrated biomedical testing equipment is expected to rise from approximately 40% in the base year 2025 to over 65% by 2033. The Compound Annual Growth Rate (CAGR) is projected to be a healthy 7.8% from 2025 to 2033. Consumer preferences are increasingly leaning towards user-friendly interfaces, comprehensive testing functionalities, and cloud-based data management solutions that facilitate compliance and reporting. Competitive dynamics are characterized by intense product development, strategic partnerships, and a growing emphasis on after-sales service and customer support. Companies are differentiating themselves through innovative features, superior accuracy, and tailored solutions for specific healthcare settings. The increasing complexity of modern medical devices, such as advanced imaging systems and robotic surgery equipment, demands equally sophisticated testing solutions, further amplifying market growth opportunities. The global market size for integrated biomedical testing equipment is estimated to reach approximately $3.5 billion by 2025, with projections to exceed $6.0 billion by 2033.
Dominant Regions & Segments in Integrated Biomedical Testing Equipment
North America currently dominates the integrated biomedical testing equipment market, driven by its advanced healthcare infrastructure, high adoption rates of sophisticated medical technologies, and a strong emphasis on patient safety and regulatory compliance. The United States, in particular, represents a significant market due to its large installed base of medical equipment and continuous investment in healthcare technology upgrades. Key drivers for this dominance include robust government funding for healthcare research and development, favorable reimbursement policies for advanced medical services, and the presence of leading medical device manufacturers and healthcare providers.
Application Segments:
- Hospitals: Represent the largest and most crucial segment due to the sheer volume of medical equipment requiring regular testing and calibration. The increasing complexity of hospital equipment, coupled with the need to ensure patient safety and comply with stringent accreditation standards, fuels demand for integrated biomedical testing solutions. Hospitals account for an estimated 60% of the total market share in 2025.
- Clinics: A growing segment, driven by the expansion of outpatient services and the increasing sophistication of equipment used in specialized clinics. Clinics are investing in reliable testing equipment to maintain device performance and patient care standards. This segment is projected to grow at a CAGR of 8.5% during the forecast period.
- Others: This segment includes research institutions, diagnostic laboratories, and medical device manufacturers, which also represent significant end-users for integrated biomedical testing equipment. Their demand is driven by R&D activities, quality control processes, and product development.
Type Segments:
- Infusion Pump Analyzers: Crucial for ensuring accurate drug delivery, these analyzers are in high demand, particularly in hospitals and critical care units. The increasing use of smart infusion pumps with advanced features further drives the need for sophisticated testing.
- Defibrillator Analyzers: Essential for maintaining the readiness of life-saving equipment, these analyzers are vital for hospitals and emergency medical services. The growing focus on cardiac care and public access defibrillation programs contributes to market growth.
- Electrosurgery Analyzers: Critical for the safe and effective operation of electrosurgical units used in a wide range of surgical procedures. The increasing number of minimally invasive surgeries boosts the demand for these analyzers.
- Gas Flow Analyzers: Used to calibrate and test medical gas delivery systems, ensuring accurate administration of anesthetic and respiratory gases. Their application extends to ventilators and anesthesia machines.
- Vital Sign Simulators: Indispensable for training healthcare professionals and testing patient monitoring systems, vital sign simulators are widely used in hospitals, educational institutions, and simulation centers.
- Electrical Safety Analyzers: A fundamental testing tool for ensuring the electrical safety of all medical equipment, preventing patient and staff harm. The broad applicability across all medical devices makes this a consistently high-demand segment.
Europe and Asia Pacific are emerging as significant growth regions, driven by increasing healthcare investments, expanding medical tourism, and a growing awareness of the importance of medical equipment maintenance and safety.
Integrated Biomedical Testing Equipment Product Innovations
Product innovations in the integrated biomedical testing equipment market are characterized by the development of multi-functional devices offering comprehensive testing capabilities from a single platform. Advanced features include real-time data acquisition, wireless connectivity for remote monitoring and cloud-based data management, AI-driven predictive maintenance insights, and enhanced automation for faster and more accurate testing. These innovations provide significant competitive advantages by reducing testing time, improving diagnostic precision, and ensuring compliance with evolving regulatory standards. The focus is on creating intuitive user interfaces and developing equipment compatible with a wider range of medical devices, including the latest generation of smart and connected healthcare technologies.
Integrated Biomedical Testing Equipment Report Scope & Segmentation Analysis
This report meticulously analyzes the integrated biomedical testing equipment market across key segmentations to provide in-depth insights into growth trajectories and market dynamics.
Application Segmentation:
- Hospitals: This segment, representing over 60% of the market share in 2025, is characterized by its high volume of diverse medical equipment and stringent regulatory demands, driving consistent demand for comprehensive testing solutions. Growth projections indicate a steady expansion driven by hospital infrastructure development globally.
- Clinics: Expected to exhibit a robust CAGR of approximately 8.5%, clinics are increasingly adopting integrated testing solutions as they expand their service offerings and invest in advanced medical equipment. The focus here is on efficient, cost-effective testing for a specialized range of devices.
- Others: This segment, encompassing research institutions and diagnostic laboratories, presents niche growth opportunities driven by R&D activities and quality assurance protocols. Market dynamics are influenced by technological advancements and the need for specialized testing capabilities.
Type Segmentation:
- Infusion Pump Analyzers: This segment is projected to grow steadily due to the critical role of infusion pumps in patient care and the ongoing advancements in infusion therapy technology.
- Defibrillator Analyzers: Demand is sustained by the essential nature of defibrillators in emergency medicine and the need for regular verification of their functionality.
- Electrosurgery Analyzers: The increasing adoption of minimally invasive surgical techniques fuels the demand for reliable electrosurgery equipment, thereby driving the market for its testing solutions.
- Gas Flow Analyzers: Essential for respiratory care and anesthesia, this segment sees consistent demand driven by the widespread use of ventilators and gas delivery systems.
- Vital Sign Simulators: Critical for training and equipment validation, these simulators are integral to medical education and clinical practice, ensuring the accuracy of patient monitoring.
- Electrical Safety Analyzers: Given their fundamental importance for all medical devices, electrical safety analyzers represent a consistently large and stable segment within the market.
Key Drivers of Integrated Biomedical Testing Equipment Growth
The integrated biomedical testing equipment market is propelled by several critical drivers. The escalating global demand for advanced and reliable healthcare services, coupled with an aging population, necessitates continuous investment in sophisticated medical equipment and its rigorous maintenance. Stringent regulatory frameworks worldwide, such as those enforced by the FDA and EMA, mandate regular testing and calibration of medical devices to ensure patient safety and efficacy, directly boosting the demand for testing equipment. Technological advancements, including the integration of AI, IoT, and cloud computing, are leading to the development of more accurate, efficient, and automated testing solutions, appealing to healthcare providers seeking to optimize operations. Furthermore, the increasing complexity of modern medical devices, from diagnostic imaging to robotic surgery systems, requires equally advanced testing capabilities, creating a consistent need for specialized integrated equipment.
Challenges in the Integrated Biomedical Testing Equipment Sector
Despite strong growth prospects, the integrated biomedical testing equipment sector faces several challenges. The high initial cost of advanced integrated testing equipment can be a significant barrier for smaller healthcare facilities and clinics, particularly in emerging economies. Evolving regulatory landscapes and the need for continuous updates to testing protocols to match new medical device technologies can create compliance complexities and necessitate ongoing investment in software and hardware upgrades. Supply chain disruptions, as observed in recent global events, can impact the availability of critical components and lead to extended lead times for equipment manufacturing and delivery. Intense competition among a growing number of manufacturers also puts pressure on pricing and profit margins, requiring companies to constantly innovate and differentiate their offerings. Cybersecurity concerns associated with connected testing equipment and the sensitive patient data they may handle also pose a challenge, requiring robust security measures.
Emerging Opportunities in Integrated Biomedical Testing Equipment
Emerging opportunities in the integrated biomedical testing equipment sector are abundant, fueled by ongoing technological advancements and evolving healthcare needs. The burgeoning field of telehealth and remote patient monitoring presents a significant opportunity for integrated testing equipment that can facilitate remote diagnostics and calibration, enabling healthcare providers to manage equipment performance without physical presence. The increasing adoption of AI and machine learning in healthcare offers scope for developing predictive maintenance capabilities within testing equipment, allowing for proactive identification and resolution of potential device failures. The growing focus on personalized medicine and the development of highly specialized medical devices create a demand for tailored testing solutions that can accurately assess the performance of these advanced instruments. Furthermore, the expansion of healthcare infrastructure in emerging economies in Asia Pacific and Africa presents substantial untapped market potential for integrated biomedical testing equipment.
Leading Players in the Integrated Biomedical Testing Equipment Market
- Fluke Corporation
- Gossen Metrawatt GmbH
- BC Group International
- Datrend Systems
- Netech Corporation
- ISimulate
- TSI Incorporated
- EKU Elektronik GmbH
- Pronk Technologies
- WHALETEQ
- Dynasthetics
- Pigeon Medical
- HE Instruments
Key Developments in Integrated Biomedical Testing Equipment Industry
- 2024 January: Fluke Corporation launched its new generation of multi-parameter patient simulator, enhancing training capabilities for healthcare professionals.
- 2023 November: Gossen Metrawatt GmbH introduced an advanced electrical safety analyzer with integrated wireless connectivity for seamless data management.
- 2023 July: BC Group International announced a strategic partnership with a leading AI firm to integrate machine learning into its upcoming defibrillator analyzers.
- 2023 March: Datrend Systems released a comprehensive software update for its electrosurgery analyzers, improving compliance reporting features.
- 2022 December: Netech Corporation expanded its product portfolio with the acquisition of a specialized gas flow analyzer manufacturer.
Future Outlook for Integrated Biomedical Testing Equipment Market
The future outlook for the integrated biomedical testing equipment market is highly promising, projected for substantial growth driven by continuous technological innovation and expanding global healthcare needs. The increasing integration of IoT and AI in medical devices will necessitate equally sophisticated and intelligent testing solutions, creating opportunities for predictive maintenance and remote diagnostics. The growing emphasis on patient safety and regulatory compliance worldwide will further solidify the demand for high-performance testing equipment. Furthermore, the ongoing expansion of healthcare infrastructure in emerging economies, coupled with a rising awareness of the importance of medical equipment reliability, will unlock significant untapped market potential. Strategic collaborations and product diversification will be key for market players to maintain a competitive edge and capitalize on the evolving landscape of healthcare technology.
Integrated Biomedical Testing Equipment Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Others
-
2. Types
- 2.1. Infusion Pump Analyzers
- 2.2. Defibrillator Analyzers
- 2.3. Electrosurgery Analyzers
- 2.4. Gas Flow Analyzers
- 2.5. Vital Sign Simulators
- 2.6. Electrical Safety Analyzers
- 2.7. Others
Integrated Biomedical Testing Equipment Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
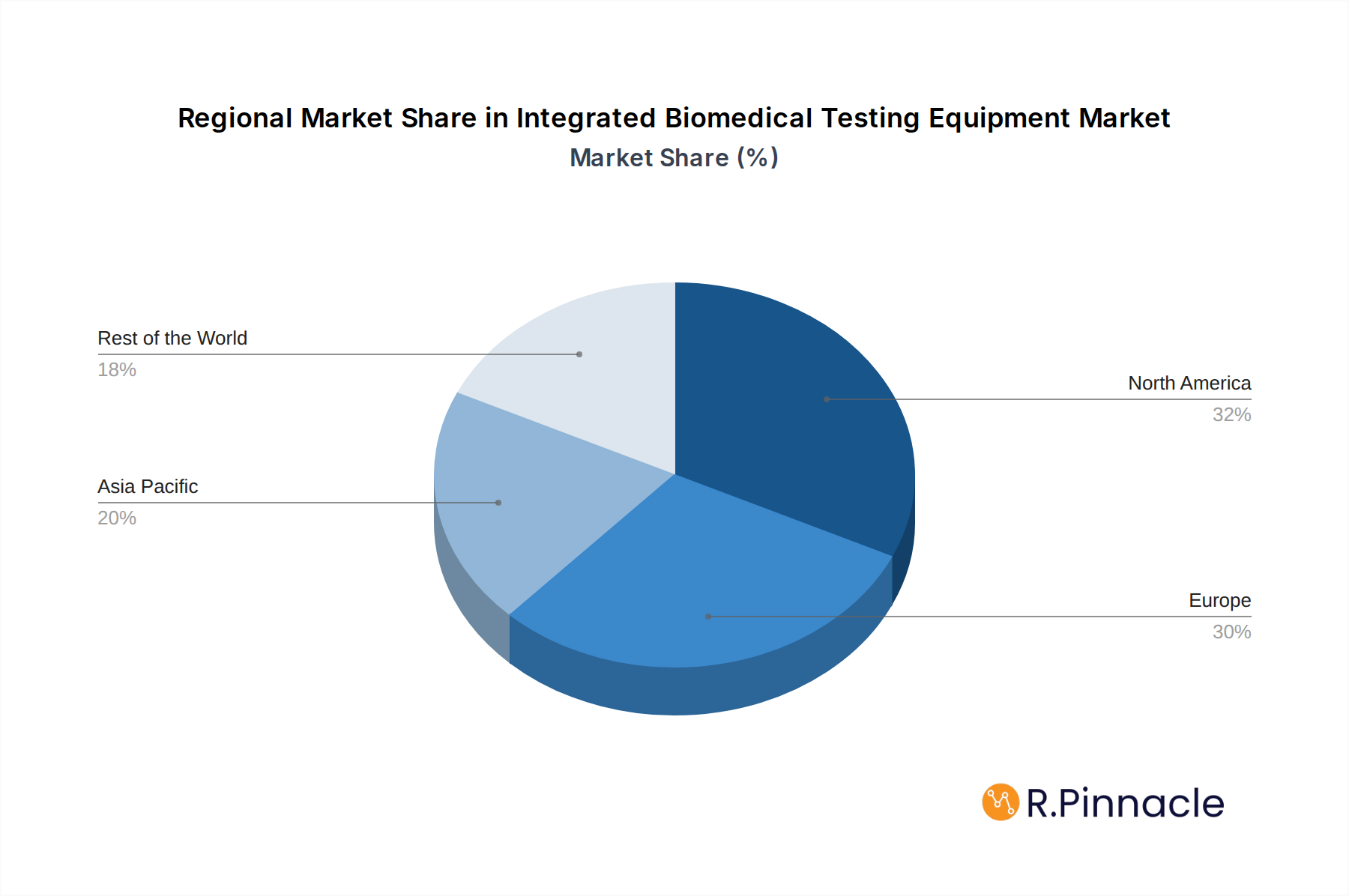
Integrated Biomedical Testing Equipment Regional Market Share

Geographic Coverage of Integrated Biomedical Testing Equipment
Integrated Biomedical Testing Equipment REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Infusion Pump Analyzers
- 5.2.2. Defibrillator Analyzers
- 5.2.3. Electrosurgery Analyzers
- 5.2.4. Gas Flow Analyzers
- 5.2.5. Vital Sign Simulators
- 5.2.6. Electrical Safety Analyzers
- 5.2.7. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Infusion Pump Analyzers
- 6.2.2. Defibrillator Analyzers
- 6.2.3. Electrosurgery Analyzers
- 6.2.4. Gas Flow Analyzers
- 6.2.5. Vital Sign Simulators
- 6.2.6. Electrical Safety Analyzers
- 6.2.7. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Infusion Pump Analyzers
- 7.2.2. Defibrillator Analyzers
- 7.2.3. Electrosurgery Analyzers
- 7.2.4. Gas Flow Analyzers
- 7.2.5. Vital Sign Simulators
- 7.2.6. Electrical Safety Analyzers
- 7.2.7. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Infusion Pump Analyzers
- 8.2.2. Defibrillator Analyzers
- 8.2.3. Electrosurgery Analyzers
- 8.2.4. Gas Flow Analyzers
- 8.2.5. Vital Sign Simulators
- 8.2.6. Electrical Safety Analyzers
- 8.2.7. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Infusion Pump Analyzers
- 9.2.2. Defibrillator Analyzers
- 9.2.3. Electrosurgery Analyzers
- 9.2.4. Gas Flow Analyzers
- 9.2.5. Vital Sign Simulators
- 9.2.6. Electrical Safety Analyzers
- 9.2.7. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Infusion Pump Analyzers
- 10.2.2. Defibrillator Analyzers
- 10.2.3. Electrosurgery Analyzers
- 10.2.4. Gas Flow Analyzers
- 10.2.5. Vital Sign Simulators
- 10.2.6. Electrical Safety Analyzers
- 10.2.7. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Integrated Biomedical Testing Equipment Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospitals
- 11.1.2. Clinics
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Infusion Pump Analyzers
- 11.2.2. Defibrillator Analyzers
- 11.2.3. Electrosurgery Analyzers
- 11.2.4. Gas Flow Analyzers
- 11.2.5. Vital Sign Simulators
- 11.2.6. Electrical Safety Analyzers
- 11.2.7. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Fluke Corporation
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Gossen Metrawatt GmbH
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 BC Group International
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Datrend Systems
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Netech Corporation
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 ISimulate
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 TSI Incorporated
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 EKU Elektronik GmbH
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Pronk Technologies
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 WHALETEQ
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Dynasthetics
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Pigeon Medical
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 HE Instruments
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.1 Fluke Corporation
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Integrated Biomedical Testing Equipment Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Integrated Biomedical Testing Equipment Revenue (million), by Application 2025 & 2033
- Figure 3: North America Integrated Biomedical Testing Equipment Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Integrated Biomedical Testing Equipment Revenue (million), by Types 2025 & 2033
- Figure 5: North America Integrated Biomedical Testing Equipment Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Integrated Biomedical Testing Equipment Revenue (million), by Country 2025 & 2033
- Figure 7: North America Integrated Biomedical Testing Equipment Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Integrated Biomedical Testing Equipment Revenue (million), by Application 2025 & 2033
- Figure 9: South America Integrated Biomedical Testing Equipment Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Integrated Biomedical Testing Equipment Revenue (million), by Types 2025 & 2033
- Figure 11: South America Integrated Biomedical Testing Equipment Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Integrated Biomedical Testing Equipment Revenue (million), by Country 2025 & 2033
- Figure 13: South America Integrated Biomedical Testing Equipment Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Integrated Biomedical Testing Equipment Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Integrated Biomedical Testing Equipment Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Integrated Biomedical Testing Equipment Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Integrated Biomedical Testing Equipment Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Integrated Biomedical Testing Equipment Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Integrated Biomedical Testing Equipment Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Integrated Biomedical Testing Equipment Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Integrated Biomedical Testing Equipment Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Integrated Biomedical Testing Equipment Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Integrated Biomedical Testing Equipment Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Integrated Biomedical Testing Equipment Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Integrated Biomedical Testing Equipment Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Integrated Biomedical Testing Equipment Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Integrated Biomedical Testing Equipment Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Integrated Biomedical Testing Equipment Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Integrated Biomedical Testing Equipment Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Integrated Biomedical Testing Equipment Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Integrated Biomedical Testing Equipment Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Integrated Biomedical Testing Equipment Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Integrated Biomedical Testing Equipment Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Integrated Biomedical Testing Equipment?
The projected CAGR is approximately 4.6%.
2. Which companies are prominent players in the Integrated Biomedical Testing Equipment?
Key companies in the market include Fluke Corporation, Gossen Metrawatt GmbH, BC Group International, Datrend Systems, Netech Corporation, ISimulate, TSI Incorporated, EKU Elektronik GmbH, Pronk Technologies, WHALETEQ, Dynasthetics, Pigeon Medical, HE Instruments.
3. What are the main segments of the Integrated Biomedical Testing Equipment?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 171 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Integrated Biomedical Testing Equipment," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Integrated Biomedical Testing Equipment report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Integrated Biomedical Testing Equipment?
To stay informed about further developments, trends, and reports in the Integrated Biomedical Testing Equipment, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


