Key Insights
The global IV training device market is poised for significant expansion, projected to reach USD 5.48 billion in 2025 and grow at a robust CAGR of 9.2% through 2033. This surge is primarily driven by the escalating demand for skilled healthcare professionals in an era of increasing medical complexity and the growing emphasis on patient safety. Hospitals, as the largest application segment, are investing heavily in advanced simulation tools to equip their nursing staff and medical technicians with proficient intravenous insertion skills. Medical schools are also recognizing the indispensable role of realistic simulation in their curriculum, enhancing the training of future healthcare providers. The market is witnessing a substantial uptake of both wearable and non-wearable IV training devices, with manufacturers innovating to offer more lifelike anatomical models, advanced haptic feedback, and digital integration for enhanced learning outcomes and procedural assessment. This evolution directly addresses the need for hands-on experience in a safe, controlled environment, reducing the learning curve and improving patient care standards.
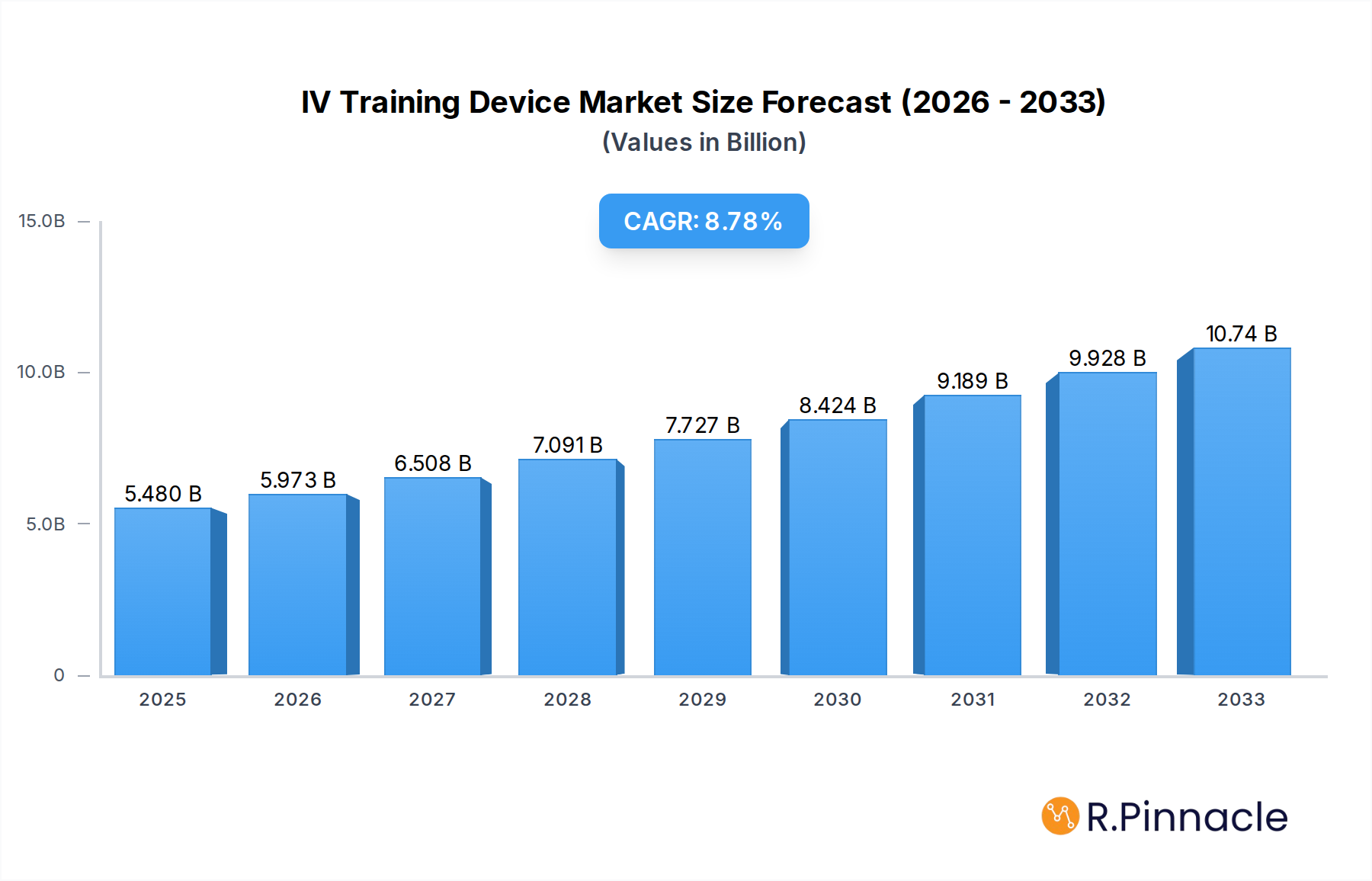
IV Training Device Market Size (In Billion)

The growth trajectory of the IV training device market is further bolstered by technological advancements and a rising awareness of their cost-effectiveness in the long run. While sophisticated devices may involve an initial investment, their ability to minimize errors, reduce material waste associated with actual patient practice, and improve procedural success rates makes them a strategic imperative for healthcare institutions. Emerging economies, with their expanding healthcare infrastructure and a burgeoning patient population, represent a key growth frontier. Companies are focusing on developing accessible and affordable training solutions to cater to these markets. Despite the strong growth outlook, challenges such as the high initial cost of some advanced simulation systems and the need for standardized training protocols across different regions could pose moderate restraints. Nevertheless, the overarching trend favors a substantial and sustained market expansion driven by the critical need for effective IV therapy training.
IV Training Device Company Market Share

IV Training Device Market Structure & Innovation Trends
The global IV Training Device market exhibits a moderate to high concentration, driven by a few dominant players such as Laerdal Medical, Gaumard, and 3B Scientific, who collectively hold an estimated 70% market share based on 2025 revenue projections. Innovation is a critical driver, with companies investing billions in R&D to develop more realistic and technologically advanced simulation devices. Key innovation trends include the integration of Artificial Intelligence (AI) for personalized feedback, haptic technology for enhanced realism, and augmented reality (AR) overlays for immersive training experiences. Regulatory frameworks, primarily focused on patient safety and educational efficacy, are continuously evolving, influencing device design and validation processes. Product substitutes, while limited in the advanced simulation space, include basic anatomical models and video-based training, though they lack the hands-on, real-time feedback crucial for proficiency. End-user demographics are primarily healthcare professionals, medical students, and nursing educators, with a growing segment of home healthcare providers seeking to enhance their skills. Mergers and Acquisitions (M&A) activity is moderate, with strategic acquisitions by larger players aiming to expand their product portfolios and market reach. For instance, a hypothetical M&A deal involving a wearable IV training technology company and a simulation software provider was valued at approximately $500 million in 2024, underscoring the consolidation trend.
IV Training Device Market Dynamics & Trends
The IV Training Device market is poised for significant growth, projected to experience a Compound Annual Growth Rate (CAGR) of XX% from 2025 to 2033. This expansion is propelled by several key market growth drivers. A primary accelerator is the increasing demand for skilled healthcare professionals globally, driven by aging populations and the rise of chronic diseases, necessitating continuous and effective training. The growing emphasis on patient safety and the reduction of medical errors further fuels the adoption of advanced simulation technologies. Technological disruptions are playing a pivotal role, with innovations in simulation fidelity, haptic feedback, and AI-driven personalized learning modules enhancing the efficacy and realism of IV training. For example, the integration of sophisticated vein visualization technologies and variable blood flow simulation are becoming standard features. Consumer preferences are shifting towards more cost-effective, accessible, and standardized training solutions that can be deployed across various educational settings. The ability of these devices to provide repeatable, objective assessments of competency is highly valued by institutions. Competitive dynamics are characterized by intense innovation and strategic partnerships, with companies vying to capture market share through superior product features and comprehensive training solutions. Market penetration is steadily increasing, particularly in developed economies, with a growing awareness of the benefits of simulation-based medical education. Emerging economies are also showing increased adoption as healthcare infrastructure and training budgets expand. The market penetration rate for advanced IV training devices is estimated to reach XX% by 2027.
Dominant Regions & Segments in IV Training Device
North America currently dominates the IV Training Device market, driven by robust healthcare expenditure, a high density of medical institutions, and a strong emphasis on continuous professional development. Within North America, the United States stands out as the leading country, with substantial investment in medical education and simulation technology.
- Application: Hospital: Hospitals represent the largest application segment, accounting for an estimated XX% of the market share in 2025. This dominance is attributed to the continuous need for training new staff, upskilling existing personnel, and maintaining competency in essential procedures like intravenous cannulation. Hospitals also utilize these devices for in-service training programs and patient education simulations.
- Application: Medical School: Medical schools constitute a significant and growing segment, driven by the increasing number of medical programs and the integration of simulation-based learning into core curricula. These institutions leverage IV training devices to provide foundational skills training in a safe and controlled environment, preparing students for clinical rotations.
- Application: Other: This segment, including nursing schools, paramedic training programs, and international aid organizations, is experiencing steady growth due to the widespread need for IV insertion skills across various healthcare professions.
- Types: Wearable: Wearable IV training devices are witnessing rapid adoption. Their portability and ability to simulate realistic venous access scenarios on anatomical models or even directly on users (for advanced applications) offer unparalleled convenience and engagement, contributing to an estimated XX% market share within the types segment.
- Types: Non-wearable: Non-wearable devices, such as benchtop simulators and manikins, remain a staple in educational institutions, offering durable and comprehensive training platforms. These devices often incorporate advanced features for vein palpation, injection, and infusion practice, holding a substantial XX% market share.
The economic policies in North America, such as government funding for healthcare education and research initiatives, further bolster the market. Advanced infrastructure for technology adoption and a skilled workforce capable of developing and utilizing these complex devices also contribute to regional dominance.
IV Training Device Product Innovations
Product innovations in the IV Training Device market are centered on enhancing realism and educational efficacy. Companies are integrating advanced haptic feedback systems to mimic the tactile sensation of vein palpation and needle insertion, alongside sophisticated fluid dynamics simulation for realistic blood flow. The incorporation of AI-powered analytics provides objective performance feedback, identifying areas for improvement and personalizing training pathways. Augmented Reality (AR) integration offers immersive learning experiences, overlaying virtual veins and anatomical structures onto physical models. These advancements provide competitive advantages by offering more effective skill development, reducing training time, and improving procedural accuracy, thereby meeting the evolving demands of the healthcare education landscape.
Report Scope & Segmentation Analysis
This report provides a comprehensive analysis of the IV Training Device market, covering key segments crucial for understanding market dynamics and growth potential.
- Application:
- Hospital: This segment, representing a substantial portion of the market, is driven by the ongoing need for clinical skill development and patient safety initiatives. Projected to grow at a CAGR of XX%, hospitals are key adopters of advanced simulation technologies for both initial and recurrent training.
- Medical School: Medical schools are increasingly integrating simulation-based training into their curricula, leading to consistent growth. This segment is expected to witness a CAGR of XX%, reflecting the growing recognition of simulation's role in producing competent future clinicians.
- Other: This diverse segment, encompassing nursing schools, paramedic programs, and other allied health professions, is projected to grow at a CAGR of XX%. The broad applicability of IV insertion skills across the healthcare spectrum fuels demand in this segment.
- Types:
- Wearable: The wearable segment is experiencing dynamic growth due to its portability and hands-on simulation capabilities. With a projected CAGR of XX%, these devices offer innovative training solutions for a variety of settings.
- Non-wearable: Non-wearable devices, including robust manikins and benchtop simulators, continue to hold a significant market share and are expected to grow at a CAGR of XX%. Their durability and comprehensive simulation features make them a cornerstone of many training programs.
Key Drivers of IV Training Device Growth
The growth of the IV Training Device market is propelled by several interconnected factors. Technologically, advancements in simulation fidelity, including realistic haptic feedback and AI-driven personalized learning, are enhancing training effectiveness and user engagement. Economically, increasing healthcare expenditures globally, coupled with a rising demand for skilled healthcare professionals, are driving institutional investment in training tools. Regulatory pressures emphasizing patient safety and the reduction of medical errors mandate the use of standardized, evidence-based training methodologies, which advanced IV training devices provide. Furthermore, the expansion of telemedicine and remote learning initiatives is creating opportunities for more accessible and distributed training solutions.
Challenges in the IV Training Device Sector
Despite robust growth, the IV Training Device sector faces several challenges. High development and manufacturing costs for advanced simulation technologies can lead to substantial upfront investments for institutions, posing a barrier to adoption, particularly in resource-constrained regions. Regulatory hurdles, while aiming to ensure quality, can also lead to lengthy validation processes for new devices. Supply chain disruptions, as seen in recent global events, can impact the availability of critical components and materials, affecting production timelines and costs. Intense competition among established players and emerging innovators also creates pressure on pricing and profit margins. The cost of advanced IV training devices can range from $XXX to $X,XXX, impacting market accessibility.
Emerging Opportunities in IV Training Device
The IV Training Device market is ripe with emerging opportunities. The growing demand for home healthcare services presents a significant avenue for the development of user-friendly and portable training devices for patients and caregivers. The integration of virtual reality (VR) and augmented reality (AR) technologies offers unprecedented opportunities for creating highly immersive and interactive training environments, enhancing skill acquisition and knowledge retention. Expansion into developing economies with rapidly growing healthcare sectors and increasing investments in medical education also presents a substantial market opportunity. Furthermore, the development of specialized training devices for rare venous anatomy or specific patient populations (e.g., pediatrics, geriatrics) can cater to niche but critical training needs.
Leading Players in the IV Training Device Market
Gaumard Medical-X Ambu 3B Scientific Adam,Rouilly BT Inc Erler-Zimmer Laerdal Medical Guangdong Kangway Technology Co.,Ltd
Key Developments in IV Training Device Industry
- 2024: Launch of AI-powered IV training simulator by Medical-X, offering personalized feedback and skill assessment.
- 2023: Gaumard introduces a new wearable IV training device with advanced haptic technology, simulating vein wall resistance.
- 2023: Laerdal Medical announces a strategic partnership with a leading VR content developer to create immersive IV training modules.
- 2022: Ambu expands its product portfolio with the acquisition of a small-to-medium enterprise specializing in anatomical models for venipuncture training.
- 2021: 3B Scientific unveils a cost-effective IV training arm designed for widespread adoption in medical schools.
Future Outlook for IV Training Device Market
The future outlook for the IV Training Device market is exceptionally promising, driven by an accelerating adoption of advanced simulation technologies and an increasing global emphasis on healthcare professional competency. The continued integration of AI, VR, and AR will revolutionize training methodologies, offering highly personalized, immersive, and effective learning experiences. As healthcare systems worldwide strive to improve patient outcomes and reduce medical errors, the demand for reliable and sophisticated IV training solutions will continue to escalate. Strategic collaborations, technological advancements, and expansion into emerging markets are expected to fuel sustained growth and innovation, positioning the IV Training Device market for substantial expansion over the forecast period.
IV Training Device Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Medical School
- 1.3. Other
-
2. Types
- 2.1. Wearable
- 2.2. Non-wearable
IV Training Device Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
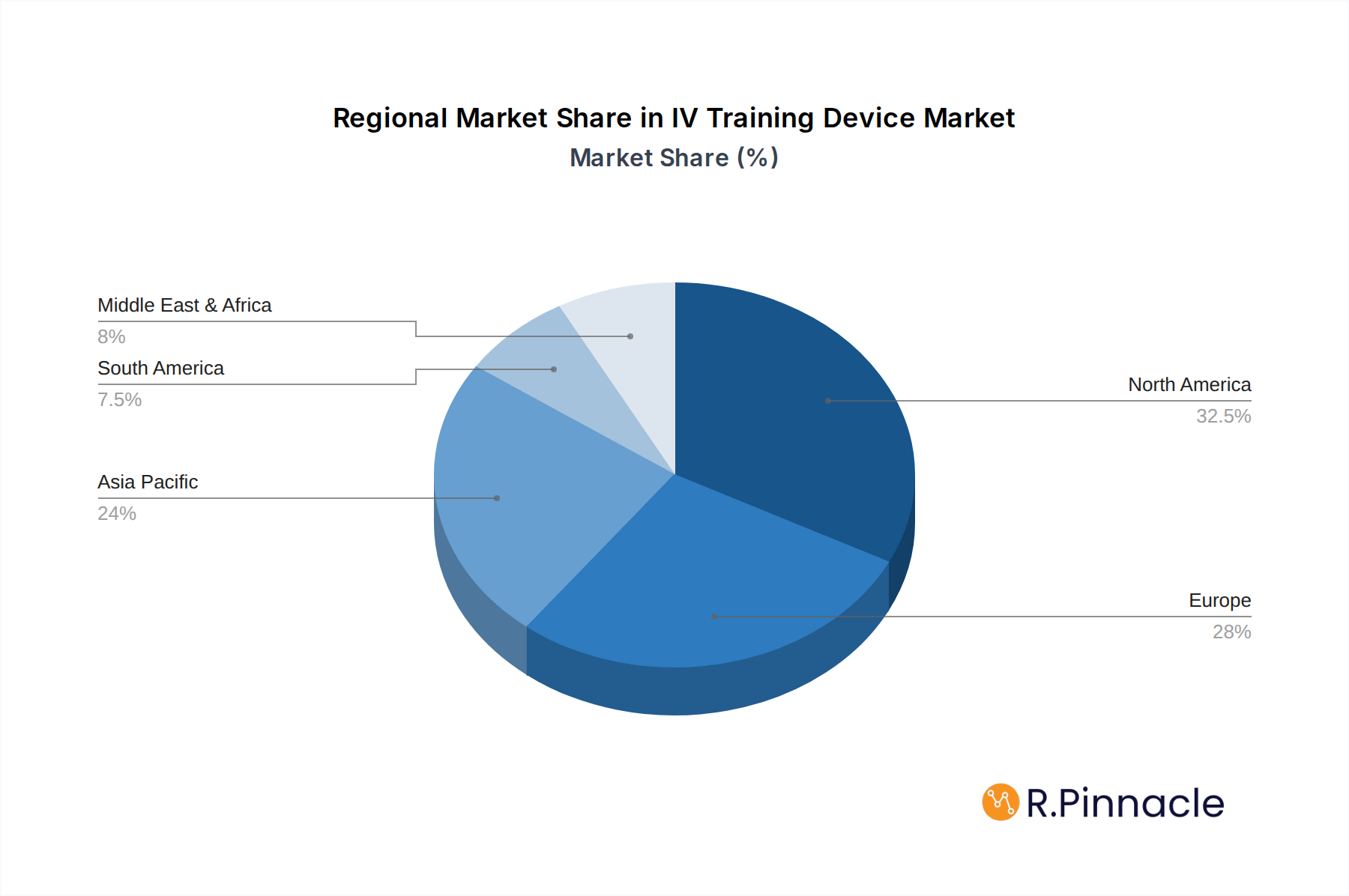
IV Training Device Regional Market Share

Geographic Coverage of IV Training Device
IV Training Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.2% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PRI Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Medical School
- 5.1.3. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Wearable
- 5.2.2. Non-wearable
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global IV Training Device Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Medical School
- 6.1.3. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Wearable
- 6.2.2. Non-wearable
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America IV Training Device Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Medical School
- 7.1.3. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Wearable
- 7.2.2. Non-wearable
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America IV Training Device Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Medical School
- 8.1.3. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Wearable
- 8.2.2. Non-wearable
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe IV Training Device Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Medical School
- 9.1.3. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Wearable
- 9.2.2. Non-wearable
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa IV Training Device Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Medical School
- 10.1.3. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Wearable
- 10.2.2. Non-wearable
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific IV Training Device Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Medical School
- 11.1.3. Other
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Wearable
- 11.2.2. Non-wearable
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Gaumard
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Medical-X
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Ambu
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 3B Scientific
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Adam
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Rouilly
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 BT Inc
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Erler-Zimmer
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Laerdal Medical
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Guangdong Kangway Technology Co.
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Ltd
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.1 Gaumard
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global IV Training Device Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global IV Training Device Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America IV Training Device Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America IV Training Device Volume (K), by Application 2025 & 2033
- Figure 5: North America IV Training Device Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America IV Training Device Volume Share (%), by Application 2025 & 2033
- Figure 7: North America IV Training Device Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America IV Training Device Volume (K), by Types 2025 & 2033
- Figure 9: North America IV Training Device Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America IV Training Device Volume Share (%), by Types 2025 & 2033
- Figure 11: North America IV Training Device Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America IV Training Device Volume (K), by Country 2025 & 2033
- Figure 13: North America IV Training Device Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America IV Training Device Volume Share (%), by Country 2025 & 2033
- Figure 15: South America IV Training Device Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America IV Training Device Volume (K), by Application 2025 & 2033
- Figure 17: South America IV Training Device Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America IV Training Device Volume Share (%), by Application 2025 & 2033
- Figure 19: South America IV Training Device Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America IV Training Device Volume (K), by Types 2025 & 2033
- Figure 21: South America IV Training Device Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America IV Training Device Volume Share (%), by Types 2025 & 2033
- Figure 23: South America IV Training Device Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America IV Training Device Volume (K), by Country 2025 & 2033
- Figure 25: South America IV Training Device Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America IV Training Device Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe IV Training Device Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe IV Training Device Volume (K), by Application 2025 & 2033
- Figure 29: Europe IV Training Device Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe IV Training Device Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe IV Training Device Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe IV Training Device Volume (K), by Types 2025 & 2033
- Figure 33: Europe IV Training Device Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe IV Training Device Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe IV Training Device Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe IV Training Device Volume (K), by Country 2025 & 2033
- Figure 37: Europe IV Training Device Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe IV Training Device Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa IV Training Device Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa IV Training Device Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa IV Training Device Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa IV Training Device Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa IV Training Device Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa IV Training Device Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa IV Training Device Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa IV Training Device Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa IV Training Device Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa IV Training Device Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa IV Training Device Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa IV Training Device Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific IV Training Device Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific IV Training Device Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific IV Training Device Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific IV Training Device Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific IV Training Device Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific IV Training Device Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific IV Training Device Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific IV Training Device Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific IV Training Device Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific IV Training Device Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific IV Training Device Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific IV Training Device Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 3: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 5: Global IV Training Device Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global IV Training Device Volume K Forecast, by Region 2020 & 2033
- Table 7: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 9: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 11: Global IV Training Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global IV Training Device Volume K Forecast, by Country 2020 & 2033
- Table 13: United States IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 21: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 23: Global IV Training Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global IV Training Device Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 33: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 35: Global IV Training Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global IV Training Device Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 57: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 59: Global IV Training Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global IV Training Device Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global IV Training Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global IV Training Device Volume K Forecast, by Application 2020 & 2033
- Table 75: Global IV Training Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global IV Training Device Volume K Forecast, by Types 2020 & 2033
- Table 77: Global IV Training Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global IV Training Device Volume K Forecast, by Country 2020 & 2033
- Table 79: China IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania IV Training Device Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific IV Training Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific IV Training Device Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the IV Training Device?
The projected CAGR is approximately 9.2%.
2. Which companies are prominent players in the IV Training Device?
Key companies in the market include Gaumard, Medical-X, Ambu, 3B Scientific, Adam, Rouilly, BT Inc, Erler-Zimmer, Laerdal Medical, Guangdong Kangway Technology Co., Ltd.
3. What are the main segments of the IV Training Device?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "IV Training Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the IV Training Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the IV Training Device?
To stay informed about further developments, trends, and reports in the IV Training Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
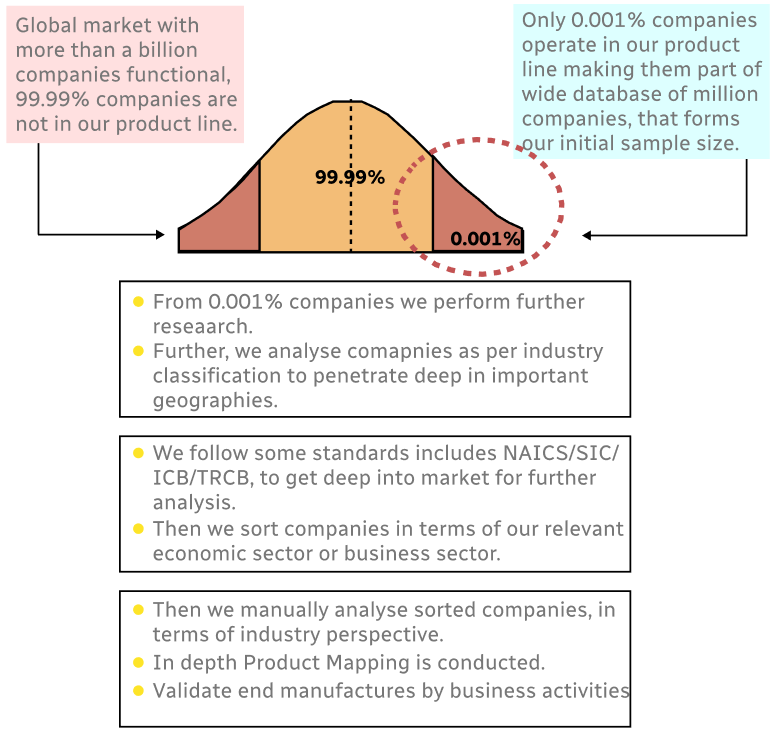
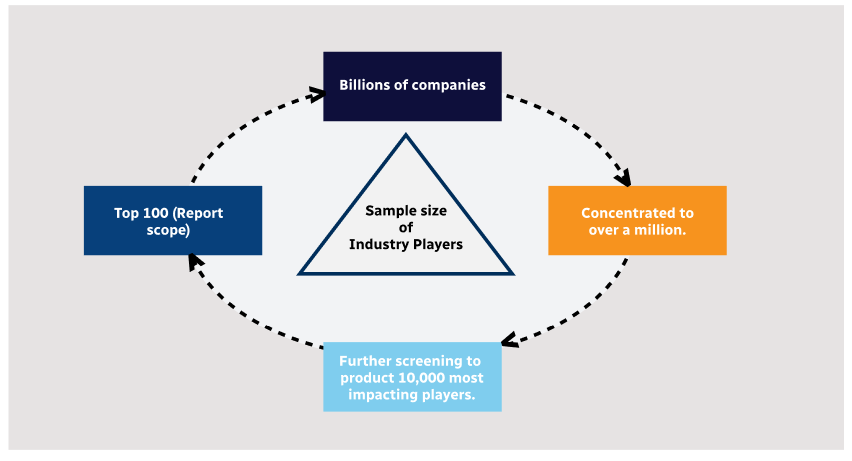
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
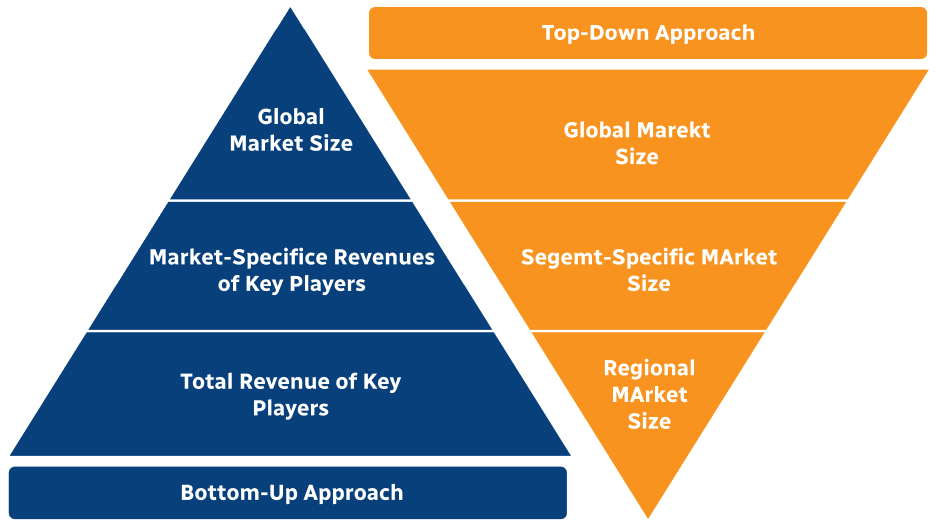
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
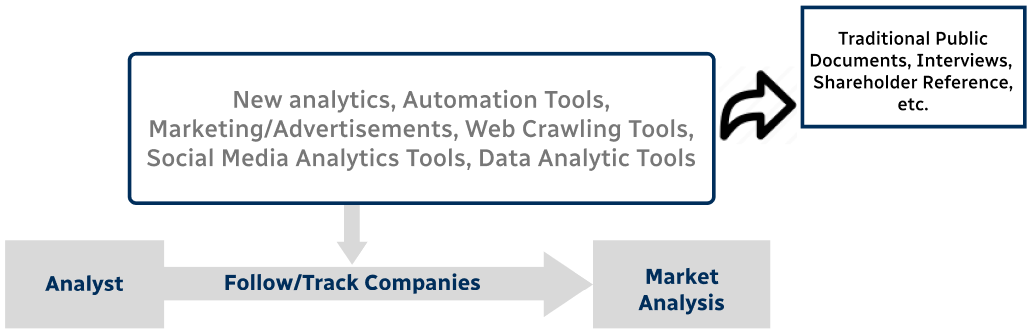
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


